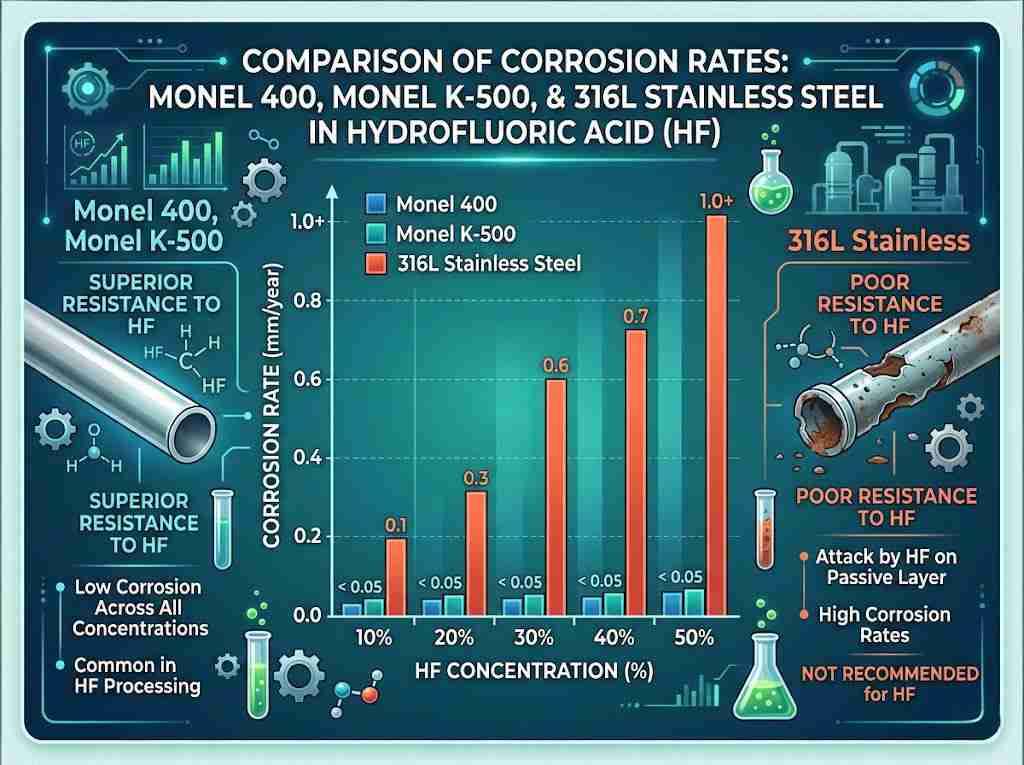
In the demanding world of chemical processing and pollution control, selecting the right material is not just about longevity; it is about safety and efficiency. For engineers and procurement officers at 28Nickel, we understand that specific environmental challenges require precise metallurgical solutions. Among the superalloys available today, Hastelloy C276 properties stand out as the benchmark for versatility and resilience in extremely corrosive environments.
This nickel-molybdenum-chromium alloy, reinforced with tungsten, is specifically designed to resist a wide range of severe chemical environments. Whether you are dealing with wet chlorine, hypochlorites, or oxidizing acids, understanding the specific Hastelloy C276 properties is the first step toward preventing catastrophic equipment failure.

The Composition Behind Hastelloy C276 Properties
To truly appreciate why this alloy performs so well, we must look at its elemental makeup. The chemical balance is what grants the unique Hastelloy C276 properties, specifically the high molybdenum content which provides resistance to localized corrosion like pitting. Furthermore, the low carbon content minimizes carbide precipitation during welding, maintaining corrosion resistance in as-welded structures.
Table 1: Nominal Chemical Composition (Weight %)
| Element | Inhalt (%) | Function |
| Nickel (Ni) | Gleichgewicht (ca. 57%) | Base matrix, stability |
| Molybdän (Mo) | 15.0 - 17.0 | Resistance to pitting & crevice corrosion |
| Chrom (Cr) | 14.5 - 16.5 | Resistance to oxidizing media |
| Eisen (Fe) | 4.0 - 7.0 | Cost efficiency & matrix support |
| Wolfram (W) | 3.0 - 4.5 | Enhanced localized corrosion resistance |
| Kobalt (Co) | 2,5 Max | Solid solution strengthening |
| Kohlenstoff (C) | 0.01 Max | Prevents intergranular corrosion |
Mechanical Hastelloy C276 Properties
While corrosion resistance is the headline feature, the mechanical strength of the material is equally vital for structural integrity. C276 retains high tensile strength and excellent ductility, making it suitable for high-pressure applications.
Unlike some distinct stainless steels that become brittle under stress, the mechanical Hastelloy C276 properties ensure the material remains tough. It can be fabricated using standard methods, though it requires specific tooling due to its work-hardening rate.
Table 2: Typical Mechanical Properties (Room Temperature)
| Eigentum | Metrischer Wert | Imperialer Wert |
| Zugfestigkeit (Ultimate) | 790 MPa (min) | 115 ksi (min) |
| Streckgrenze (0,2% Offset) | 355 MPa (min) | 52 ksi (min) |
| Dehnung | 40% | 40% |
| Härte (Rockwell B) | 100 max | 100 max |
Corrosion Resistance: The Core of Hastelloy C276 Properties
The primary reason industries turn to 28Nickel for this alloy is its legendary resistance profile. The Hastelloy C276 properties regarding corrosion are exceptionally broad.
-
Pitting and Crevice Corrosion: The high Molybdenum and Tungsten content gives C276 a high Pitting Resistance Equivalent Number (PREN). This makes it virtually immune to crevice corrosion in seawater and brine environments.
-
Stress Corrosion Cracking (SCC): Chloride-induced SCC is the downfall of many austenitic stainless steels. However, Hastelloy C276 properties include exceptional immunity to SCC, making it vital for sour gas wells and heat exchangers handling chlorides.
-
Oxidizing and Reducing Environments: The Chromium content combats oxidizing media (like nitric acid), while the Molybdenum helps in reducing environments (like hydrochloric and sulfuric acids). This “dualability” is rare.
Physical Hastelloy C276 Properties
For design engineers calculating heat transfer or weight, the physical attributes are non-negotiable data points.
-
Density: 8.89 g/cm³ (0.321 lb/in³)
-
Melting Range: 1325 – 1370°C (2415 – 2500°F)
-
Electrical Resistivity: 1.30 µΩ-m at 24°C
These physical Hastelloy C276 properties indicate a material that is dense and stable at high temperatures, although it is generally not used for service temperatures above 1250°F (677°C) if corrosion resistance is the priority, due to the formation of precipitates.
Fabrication and Welding Characteristics
A common misconception is that superalloys are impossible to weld. In reality, Hastelloy C276 properties are favorable for welding. It is compatible with GTAW (TIG), GMAW (MIG), and SMAW (Stick) processes.
Because the alloy has a low carbon content, the formation of grain-boundary precipitates in the heat-affected zone (HAZ) is suppressed. This means that for most chemical process applications, the alloy can be used in the “as-welded” condition without requiring post-weld solution heat treatment. However, machinists must be aware that C276 work-hardens rapidly; rigid setups and aggressive feed rates are recommended to prevent glazing the surface.
At 28Nickel, we see Hastelloy C276 as the “workhorse” of the superalloy family. It does not just perform; it endures. By thoroughly reviewing these Hastelloy C276 properties, from its molybdenum-rich composition to its mechanical toughness, engineers can confidently specify this material for the harshest scrubbers, ducts, and reactor vessels.
Verwandte Fragen und Antworten
Q1: How do Hastelloy C276 properties compare to Stainless Steel 316?
A: While 316 is a standard corrosion-resistant material, Hastelloy C276 properties are far superior in aggressive environments. C276 offers resistance to wet chlorine and hot concentrated acids where 316 would fail almost immediately due to pitting or stress corrosion cracking.
Q2: Is Hastelloy C276 magnetic?
A: No. One of the physical Hastelloy C276 properties is that it is non-magnetic. This makes it suitable for applications where magnetic interference must be minimized, unlike some ferritic or martensitic steel grades.
Q3: Can Hastelloy C276 be used at high temperatures?
A: Yes, but with caveats. While it has high strength, prolonged exposure to temperatures above 677°C (1250°F) can lead to the formation of precipitates which may degrade corrosion resistance. Therefore, Hastelloy C276 properties are best utilized in lower temperature, high-corrosion wet environments.