Cuando los ingenieros preguntan por Alloy 20 corrosion resistance in sulfuric acid, they are usually not looking for a generic alloy profile. They want to know whether the material will survive real process conditions: mixed acid concentration, temperature cycling, trace chlorides, stagnant zones, welds, and startup or shutdown excursions. That is exactly where alloy selection stops being a catalog exercise and becomes a corrosion engineering problem.
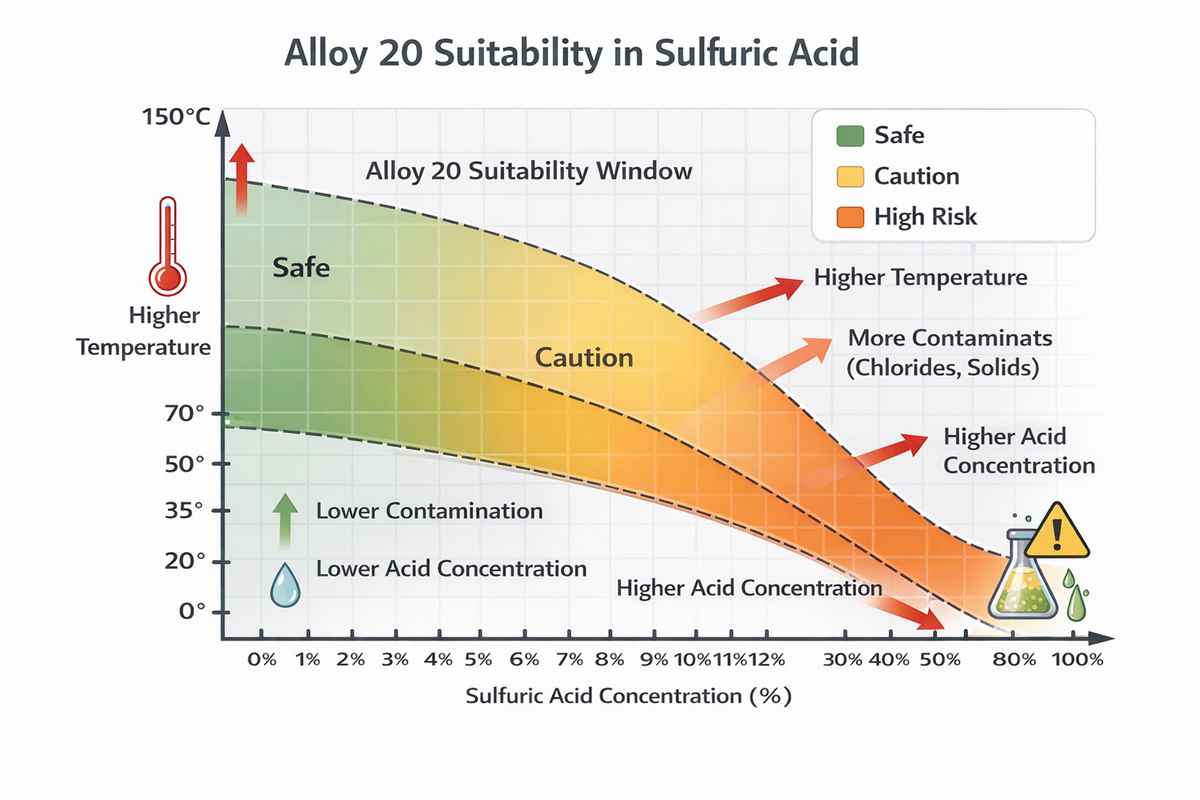
Alloy 20 was developed for sulfuric acid service, and that heritage still matters. Its balanced Ni-Cr-Mo-Cu chemistry gives it a distinct niche between stainless steels that are too easily attacked and higher-nickel alloys that may be technically excellent but commercially excessive for moderate duty. In practice, Alloy 20 corrosion resistance in sulfuric acid is best understood not as a single “good” or “bad” rating, but as a window of serviceability shaped by acid strength, oxidizing potential, temperature, contamination, flow regime, and fabrication quality.
Why Alloy 20 Performs in Sulfuric Acid Service
The reason Alloy 20 is often considered for sulfuric acid is metallurgical, not marketing. Nominally, the alloy combines high nickel with chromium, molybdenum, and copper, plus niobium stabilization. Each element contributes differently.
Nickel improves resistance in reducing media and helps the alloy avoid some of the rapid general corrosion that can limit conventional austenitic stainless steels. Chromium supports passive film formation when conditions are sufficiently oxidizing. Molybdenum adds value in complex corrosive environments, especially when localized attack becomes a concern. Copper is especially relevant here: in sulfuric acid service, copper can materially improve performance in certain concentration ranges where stainless grades such as 316L lose ground quickly.
Por ello Alloy 20 corrosion resistance in sulfuric acid is often stronger than engineers first expect when they compare it only by chromium or nickel level. The alloy was engineered for a specific corrosion problem, and its composition reflects that purpose.
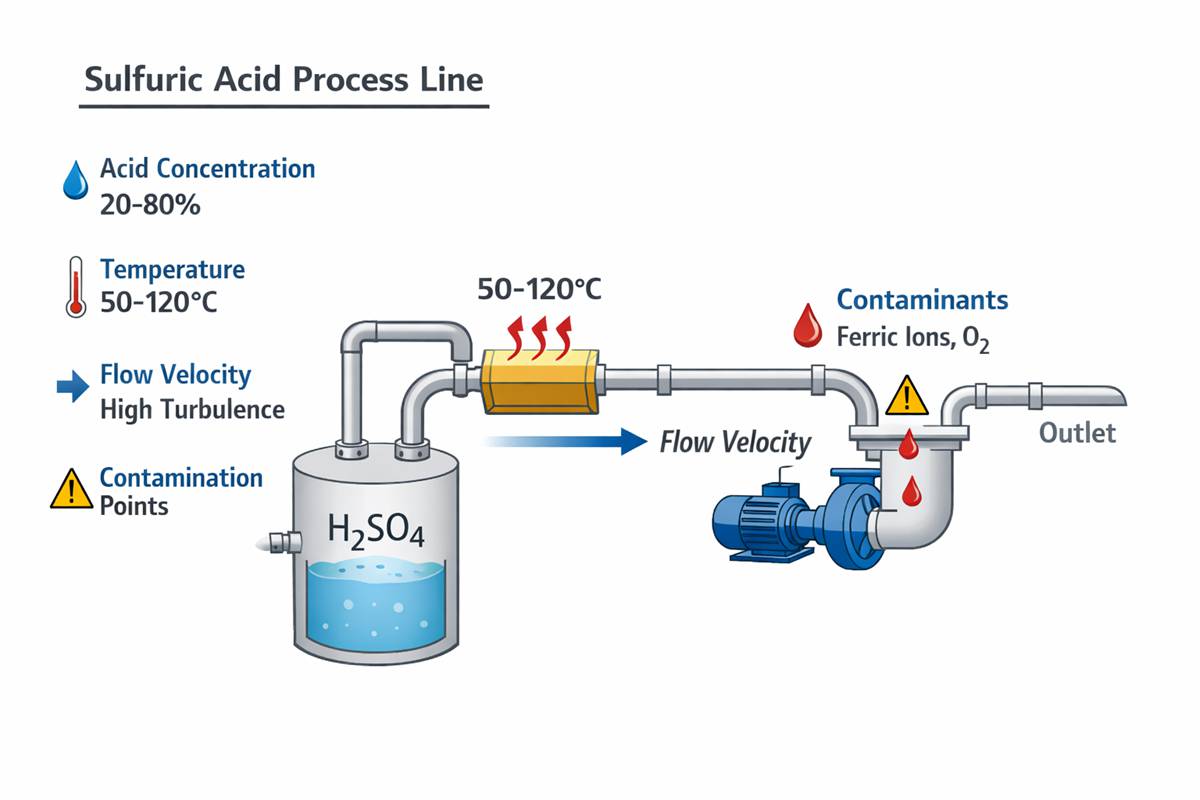
Still, there is no universal immunity. Sulfuric acid is a deceptively variable environment. A line carrying nominally the same acid concentration may behave very differently if it contains ferric ions, chlorides, entrained solids, dissolved oxygen, or reducing contaminants. A storage tank in steady ambient service is one problem. A heated reactor feed line with intermittent flow and poor drainage is another entirely.
What Controls Alloy 20 Corrosion Resistance in Sulfuric Acid?
From an engineering standpoint, the first variable is concentration. Sulfuric acid is not linear in its corrosivity. Some alloys perform acceptably in dilute acid and fail in more concentrated acid; others show the opposite trend over limited temperature bands. Therefore, Alloy 20 corrosion resistance in sulfuric acid should never be evaluated without concentration and temperature stated together.
The second variable is temperature, and it is often the one that decides the project. Many alloys that look acceptable on paper at ambient conditions lose margin rapidly as temperature rises. Corrosion rates can accelerate sharply, and localized phenomena become more difficult to predict. If your process sees hot acid, upset heating, or steam tracing near dead legs, the available corrosion allowance can disappear much faster than expected.
The third variable is contamination. Chlorides are especially important because they can shift the problem from general metal loss to pitting, crevice attack, or attack in poorly drained weld details. Oxidizing species can either improve or worsen performance depending on the chemistry balance. Iron salts, copper salts, process organics, and abrasive solids all change the effective environment. In other words, plant acid is rarely “pure acid.”
This is why experienced engineers do not discuss Alloy 20 corrosion resistance in sulfuric acid in isolation. They ask sharper questions: Is the acid aerated? Is there chloride carryover? Are there solids? Is the system continuously wetted? What is the shutdown condition? Is the weld pickled and passivated, or left as-welded? These details are not secondary. They are the service environment.
Practical Design Factors for Alloy 20 in Sulfuric Acid Systems
| Design factor | Por qué es importante | Practical engineering guidance |
|---|---|---|
| Acid concentration | Corrosion behavior changes significantly across concentration ranges | Always define exact concentration range, not just “sulfuric acid service” |
| Temperatura | Rising temperature can sharply increase corrosion rate | Review normal, upset, and cleaning temperatures separately |
| Oxidizing vs. reducing condition | Passivity and metal dissolution behavior can shift | Confirm dissolved oxygen, ferric ions, and redox condition |
| Chloride contamination | Can promote pitting and crevice corrosion | Check upstream contamination and stagnant crevice locations |
| Flow regime | Erosion-corrosion and turbulence can remove protective films | Evaluate pumps, elbows, reducers, and flashing zones |
| Weld condition | Weld oxide and poor finishing can reduce corrosion margin | Specify qualified filler, controlled heat input, and post-fabrication cleaning |
| Deposits and solids | Under-deposit attack can make a “good alloy” fail locally | Design for drainage, access, and periodic inspection |
| Startup/shutdown exposure | Condensation and concentration changes can be more severe than steady operation | Assess transient exposure, not only normal operating conditions |
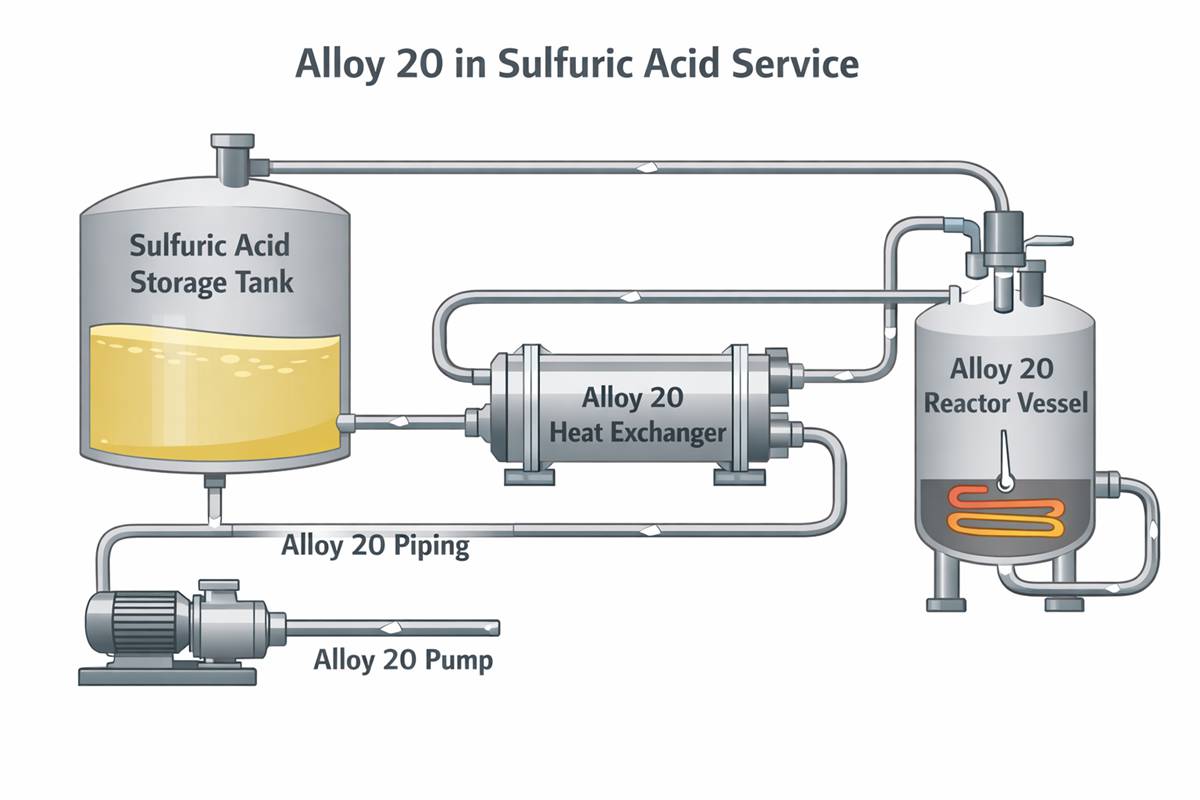
Where Alloy 20 Works Well—and Where It Does Not
In many sulfuric acid plants, chemical process units, pickling systems, and fertilizer-related services, Alloy 20 corrosion resistance in sulfuric acid makes it a rational choice when 304L or 316L would be too risky, but a much higher alloy would be difficult to justify economically. That is the practical middle ground where Alloy 20 earns its reputation.
However, engineers should be careful not to oversimplify that reputation. Alloy 20 is not the default answer for every hot sulfuric acid stream, every mixed-acid system, or every chloride-bearing unit. When temperature rises, crevices are unavoidable, or contamination is severe, the alloy may move out of its comfortable operating window. Under those conditions, more highly alloyed nickel materials may offer the reliability margin required for long campaign life.
Another common mistake is to treat published corrosion data as directly transferable to fabricated equipment. It is not. Lab data are useful for screening, but nozzle geometry, weld profile, drainability, insulation details, and maintenance practice often decide field performance. Good material can be defeated by poor design. Conversely, a disciplined design and fabrication package can make Alloy 20 corrosion resistance in sulfuric acid deliver excellent service life in applications where lesser alloys show premature attack.
For procurement teams, this means the right question is not simply, “Is Alloy 20 resistant to sulfuric acid?” The better question is, “Under our exact chemistry and temperature envelope, what corrosion mechanism is most likely, and what fabrication controls are required?” That is where material selection becomes reliable.
Ingeniería Conclusión
The real value of Alloy 20 is that it was designed with sulfuric acid in mind, and that design intent still shows in service. But Alloy 20 corrosion resistance in sulfuric acid is not a blanket guarantee. It is a performance envelope. Within the right concentration, temperature, and contamination limits, the alloy can be highly effective and cost-efficient. Outside that envelope, failure can be quiet, localized, and expensive.
For serious projects, the most defensible approach is to combine published corrosion data, plant chemistry review, fabrication requirements, and—when the duty is critical—application-specific corrosion testing. That is the level of engineering rigor that turns a material grade from a brochure choice into a dependable asset.
Preguntas y respuestas relacionadas
1. Is Alloy 20 better than 316L in sulfuric acid?
In many sulfuric acid applications, yes. Alloy 20 was specifically developed for this service and generally offers a much wider usable window than 316L. The real comparison, however, must include acid concentration, temperature, and contamination.
2. Does Alloy 20 resist sulfuric acid at high temperature?
It can, but only within a limited operating window. As temperature rises, corrosion rates may increase quickly, and the presence of chlorides, deposits, or crevices can narrow the safe range further. High-temperature duty should be checked case by case.
3. What is the biggest mistake when selecting Alloy 20 for sulfuric acid service?
Treating sulfuric acid as a single environment. Concentration, redox condition, chlorides, solids, welding condition, and shutdown exposure all affect performance. Most field failures come from underestimating those combined variables.