In the rigorous field of materials engineering, selecting the right alloy for highly corrosive environments—such as alkylation units, marine splash zones, and chemical processing facilities—leaves no room for guesswork. Standard austenitic stainless steels often suffer rapid degradation when exposed to reducing acids. To understand why certain nickel-copper matrices excel where others deteriorate catastrophically, we must fundamentally examine the monel alloy chemical composition.
In my two decades of evaluating metallurgical failures and specifying alloys for severe service conditions, I have consistently found that the secret to this material’s longevity lies not just in its primary elements, but in the precise thermodynamic balance of its micro-alloying additions. The monel alloy chemical composition is engineered to form a single-phase solid solution, granting it exceptional structural stability from sub-zero temperatures up to 480°C (900°F).

Decoding the Monel Alloy Chemical Composition Baseline
When we analyze the baseline monel alloy chemical composition, specifically focusing on the widely utilized Alloy 400 (UNS N04400), we are looking at a binary system dominated by Nickel (typically 63% minimum) and Copper (28% to 34%). This specific ratio is not arbitrary; it mirrors the natural composition found in the meteoritic nickel-copper ores from the Sudbury basin.
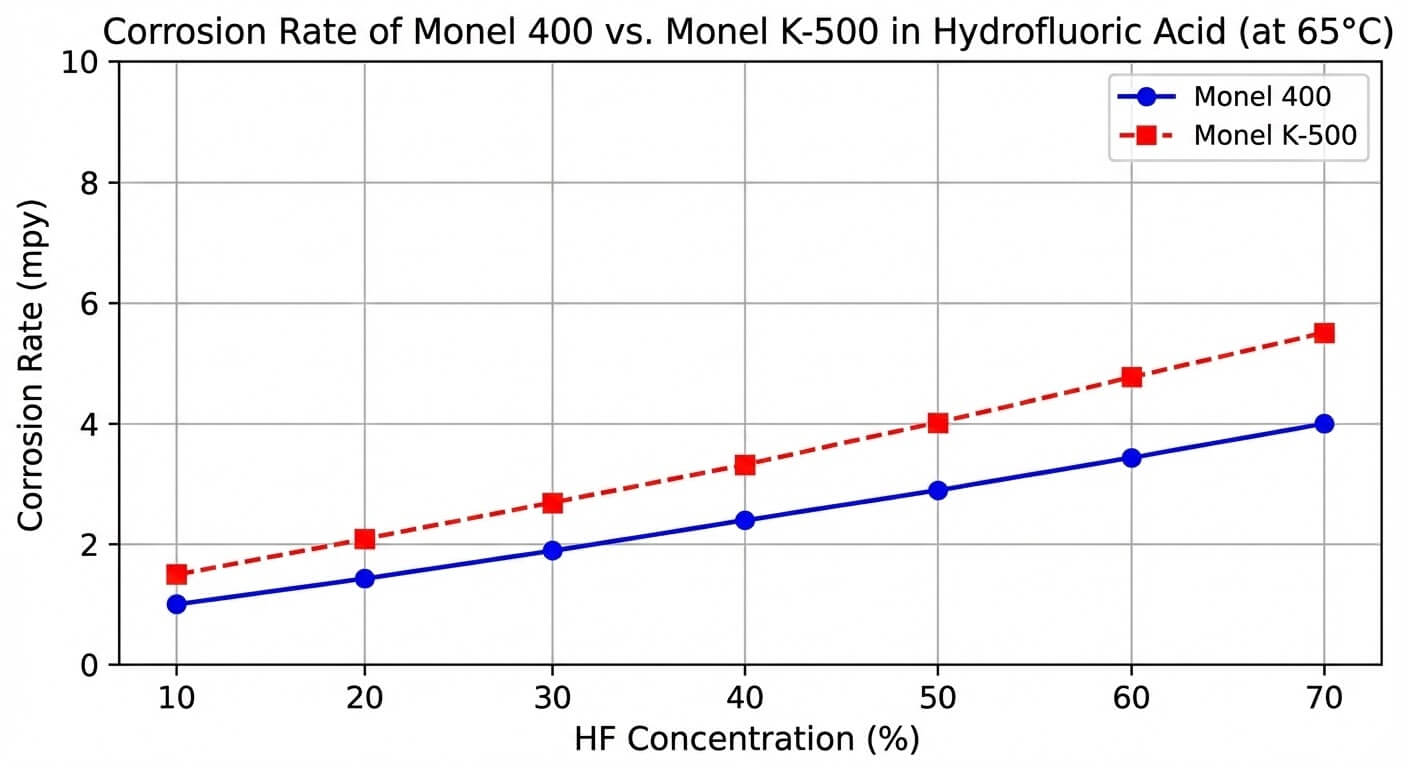
Nickel provides the noble matrix that inherently resists chloride-ion stress corrosion cracking (SCC)—a notorious failure mode for 300-series stainless steels in marine and petrochemical applications. Copper is the critical addition that enhances the alloy’s resistance to reducing environments, most notably hydrofluoric acid (HF) and sulfuric acid under non-aerated conditions. In hydrofluoric acid service, the exact monel alloy chemical composition allows the material to form a highly tenacious, protective cupric fluoride film on the surface. If the copper content drops below the specified threshold, or if oxidizing agents are introduced into the process stream, this protective film destabilizes, leading to accelerated localized attack.
Furthermore, trace elements within the monel alloy chemical composition dictate its manufacturability. Iron (up to 2.5%) and Manganese (up to 2.0%) are precisely controlled. Manganese acts as a vital deoxidizer and sulfur scavenger during the melting process, preventing hot-shortness and ensuring the material can be successfully forged and rolled without internal tearing.
Standard Monel Alloy Grade Comparison
To better understand the metallurgical variance, we must compare the elemental limits across different grades.
| Elemento | Alloy 400 (UNS N04400) | Alloy K-500 (UNS N05500) | Alloy R-405 (UNS N04405) |
| Níquel (Ni) | 63.0% min | 63.0% min | 63.0% min |
| Cobre (Cu) | 28.0 – 34.0% | 27.0 – 33.0% | 28.0 – 34.0% |
| Hierro (Fe) | 2.5% max | 2.0% max | 2.5% max |
| Manganeso (Mn) | 2.0% max | 1.5% max | 2.0% max |
| Aluminio (Al) | – | 2.3 – 3.15% | – |
| Titanio (Ti) | – | 0.35 – 0.85% | – |
| Azufre (S) | 0.024% max | 0.010% max | 0.025 – 0.060% |
Variations in Monel Alloy Chemical Composition
While Alloy 400 serves as the workhorse, modern engineering often demands higher mechanical strength without sacrificing corrosion resistance. This is where precipitation-hardenable variations come into play. By modifying the monel alloy chemical composition to include Aluminum and Titanium, metallurgists created Alloy K-500.
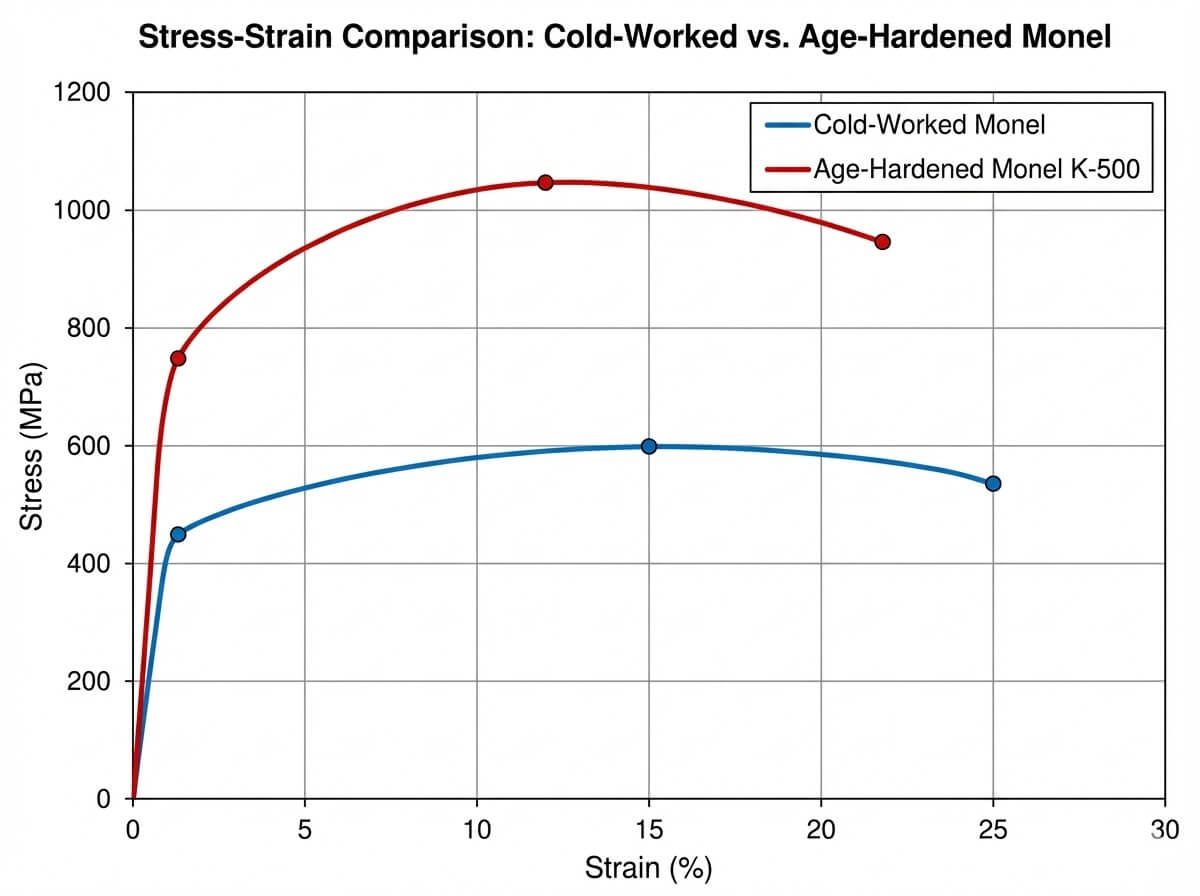
During controlled thermal aging treatments, these specific elemental additions precipitate out of the solid solution as sub-microscopic particles of Ni3(Ti, Al), known as the gamma prime phase. This phase locks the dislocation movement within the crystal lattice, effectively tripling the yield strength and doubling the tensile strength compared to the annealed baseline grade. However, engineers must be acutely aware that this modified monel alloy chemical composition requires strict adherence to heat treatment protocols; improper aging can lead to a phenomenon known as age-hardening embrittlement, particularly in hydrogen-rich environments.
Another critical variation is Alloy R-405. For applications requiring rapid, high-volume machining—such as the manufacturing of fasteners, valve stems, and precision screw machine products—a slight but vital tweak is made to the monel alloy chemical composition. By intentionally increasing the Sulfur content (up to 0.060%), nickel sulfides form within the matrix. These sulfides act as microscopic chip breakers during machining operations, significantly reducing tool wear and improving surface finish.
Engineering the Right Solution
Ultimately, specifying materials for critical infrastructure is an exercise in managing metallurgical constraints. A superficial understanding of these alloys will inevitably lead to premature equipment failure, process contamination, and catastrophic safety risks. The monel alloy chemical composition is a finely tuned metallurgical system, where every percentage point of an alloying element serves a distinct structural or protective purpose.
Whether your project involves marine engineering, chemical processing, or oil and gas extraction, ensuring the exact elemental balance is non-negotiable. If you are currently evaluating materials for an aggressive process environment, our engineering team at 28Nickel is available to provide deep-dive technical support, review your corrosion data, and help you determine the precise metallurgical specifications required for your operational success.
Preguntas y respuestas relacionadas
Q1: How does iron limit specification affect the monel alloy chemical composition?
Iron is kept strictly below 2.5% in standard grades. While it provides some solid solution strengthening, excessive iron in the monel alloy chemical composition can decrease the material’s resistance to highly reducing acids like hydrofluoric acid and increase the risk of localized corrosion pitting in stagnant seawater.
Q2: Is the monel alloy chemical composition inherently resistant to sour gas (H2S)?
Yes, up to a certain threshold. The high nickel and copper baseline in the monel alloy chemical composition offers excellent resistance to sulfide stress cracking in sour gas environments, which is why Alloy K-500 is frequently specified for downhole drill collars and pump shafts in the oil and gas industry, provided the hardness levels comply with NACE MR0175 standards.
Q3: Why does the monel alloy chemical composition fail in oxidizing environments?
The core Ni-Cu monel alloy chemical composition relies on reducing conditions to maintain its protective surface films. In the presence of strong oxidizers like nitric acid, ferric salts, or highly aerated ammonia, the copper content becomes a liability, leading to rapid dissolution of the alloy matrix. In such cases, chromium-bearing alloys (like Inconel or Hastelloy) are structurally required.