Quando gli ingegneri chiedono informazioni su Inconel 718 corrosion resistance in sulfuric acid, my first reaction is usually the same: do not answer too quickly. Alloy 718 is a very capable nickel-chromium alloy, but sulfuric acid is one of those environments where a casual “nickel alloy = safe” mindset gets people into trouble. Special Metals lists 718 as a precipitation-hardenable Ni-Cr alloy with about 50–55% nickel, 17–21% chromium, and 2.8–3.3% molybdenum, and describes it as having excellent corrosion resistance in many media. That broad statement is true, but it is not the same thing as saying 718 is a dedicated sulfuric-acid alloy.
The real engineering answer is that Inconel 718 corrosion resistance in sulfuric acid depends heavily on four variables: acid concentration, temperature, oxidizing or reducing contaminants, and whether corrosion resistance or mechanical strength is the primary design driver. Haynes notes that sulfuric acid behavior is strongly concentration- and temperature-dependent, that molybdenum is highly beneficial in pure sulfuric acid, that copper is also advantageous, and that field trials are recommended because plant chemistry rarely behaves like clean laboratory acid.
That last point matters. In real plants, sulfuric acid may carry ferric ions, chlorides, sulfates, process organics, or oxidizing species from upstream operations. Once that happens, the passive film stability and uniform corrosion rate can shift fast. So, if your question is whether Inconel 718 corrosion resistance in sulfuric acid is “good,” the answer is not yes or no. It is: good enough for what exact chemistry, what exact temperature, and what exact failure mode?
What Inconel 718 corrosion resistance in sulfuric acid really depends on
From a metallurgical standpoint, 718 was designed to balance high strength, weldability, and general corrosion resistance. Its corrosion resistance comes mainly from nickel and chromium, while its molybdenum level is modest. Special Metals states that nickel supports resistance in many inorganic and organic media, chromium helps against oxidizing media and sulfur compounds, and molybdenum contributes to pitting resistance. That is a sound foundation, but it is still different from alloys deliberately optimized for sulfuric acid duty.
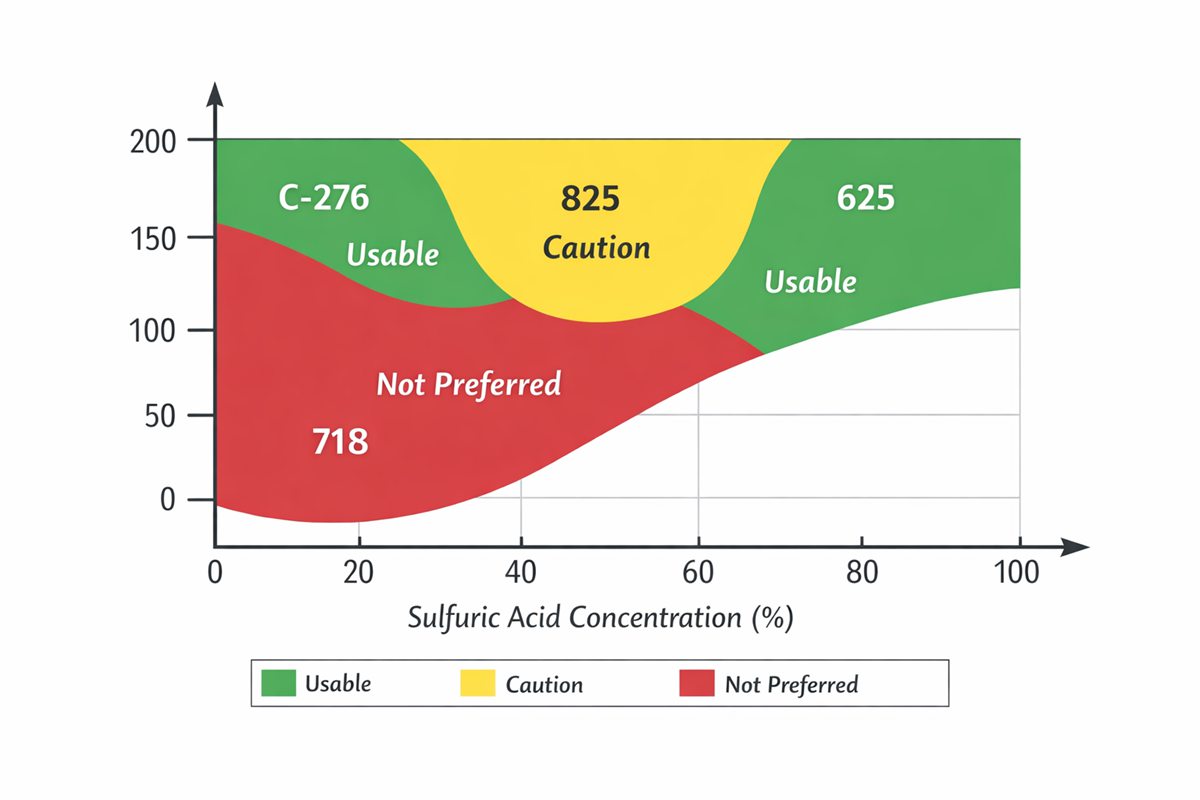
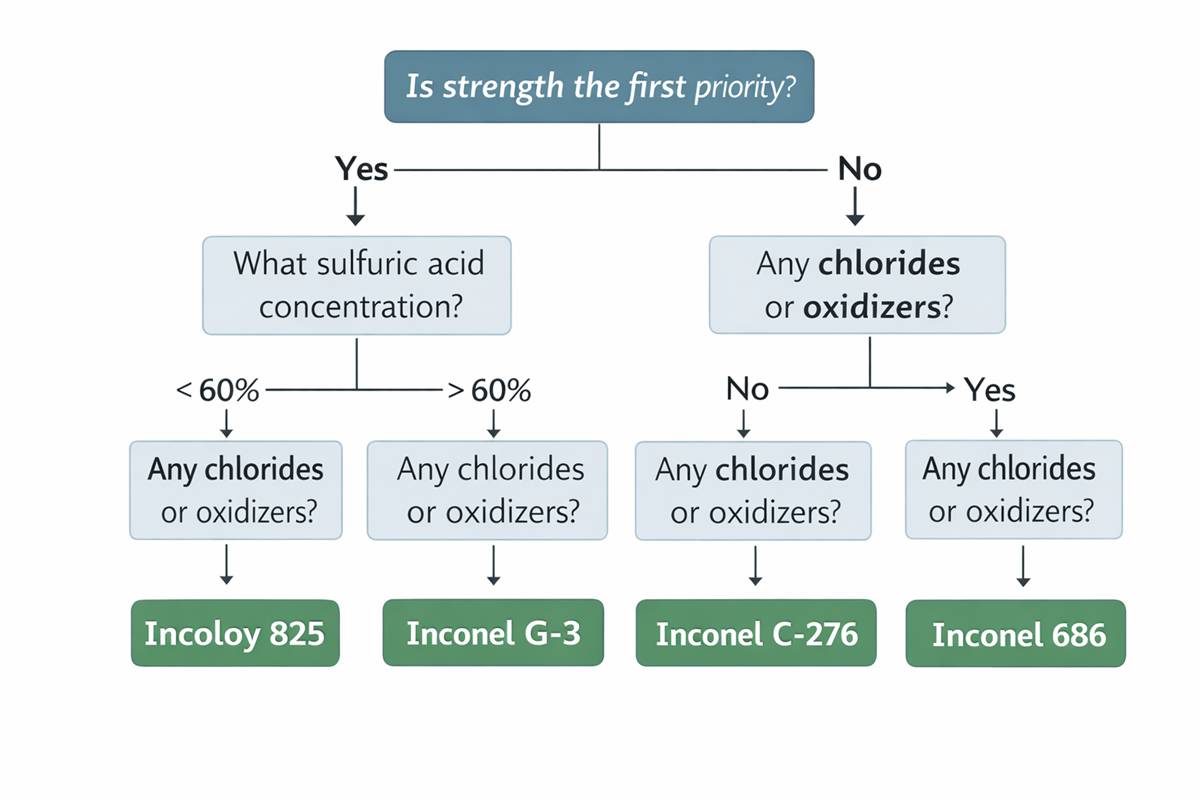
This is why experienced corrosion engineers usually compare 718 against sulfuric-acid specialists before signing off. In the same Special Metals references, INCOLOY 825 is described as having excellent resistance to sulfuric and phosphoric acids; INCONEL G-3 is explicitly used for sulfuric acid handling; C-276 is positioned for reducing and mildly oxidizing environments; and 686 is described as having excellent resistance to oxidizing, reducing, and mixed acids. Those descriptions are not marketing trivia. They reflect alloy design intent. 718, by contrast, is presented primarily as a high-strength age-hardenable alloy with corrosion resistance, not as a first-line sulfuric acid material.
The sulfuric acid ranking data in Special Metals’ aqueous corrosion handbook reinforces that point. For non-deaerated sulfuric acid, the handbook ranks 825, 686, and G-3 among the stronger performers in dilute acid, while 686 and C-276 lead in concentrated acid; 718 does not appear in that sulfuric-acid ranking set. That does not prove 718 is unusable. It does show that when sulfuric acid is the main corrosive variable, the industry normally looks first to other leghe di nichel.
Sulfuric acid alloy selection table
| Lega | Practical sulfuric-acid positioning | Why engineers choose it |
|---|---|---|
| Inconel 718 | Use with caution when sulfuric acid exposure is secondary and high strength is mandatory | Strong age-hardenable alloy for fasteners, shafts, springs, and oilfield parts where strength may outweigh pure corrosion optimization |
| Incoloy 825 | Common benchmark for intermediate sulfuric acid service | Ni-Fe-Cr-Mo-Cu design is specifically positioned for sulfuric acid resistance |
| Inconel G-3 | Strong candidate for sulfuric and phosphoric acid systems | Higher Mo plus Cu, and explicitly associated with sulfuric acid handling |
| Inconel 625 | Better general corrosion choice than 718 when strength is still needed | Higher molybdenum and stronger reputation in severe corrosive media |
| Inconel C-276 | Standard choice for reducing to mildly oxidizing acid environments | High Mo chemistry gives it much broader acid-service credibility |
| Inconel 686 | Premium option for aggressive mixed-acid and chloride-contaminated service | Excellent resistance to oxidizing, reducing, and mixed acids |
Table note: The alloy positioning above summarizes Special Metals and Haynes chemistry and application guidance for 718, 825, G-3, 625, C-276, and 686.
When 718 still makes engineering sense
So where does Inconel 718 corrosion resistance in sulfuric acid actually make sense? In my view, 718 becomes reasonable when the component is mechanically demanding and the acid exposure is limited, intermittent, diluted, or tightly controlled. Think high-strength bolting, springs, downhole hardware, valve stems, or structural parts that cannot simply be switched to a softer corrosion alloy without redesigning the load path. Special Metals also notes 718 is produced for oilfield applications under NACE MR0175/ISO 15156 related service conditions, which supports the idea that 718 often enters corrosive service because of its strength package, not because it is the best sulfuric-acid alloy on the shelf.
I would be far more conservative for hot circulating sulfuric acid, pickling circuits, evaporators, acid recovery loops, and stagnant crevice geometries. Even in seawater, where 718 shows excellent corrosion-fatigue strength, Special Metals notes it is less resistant to crevice corrosion than 625, 625LCF, and 725. That is not sulfuric-acid data, but it is a useful reminder that 718 sits below the top tier of nickel corrosion alloys when localized corrosion margin becomes critical.
A second caution concerns contaminated acid. Special Metals reports that oxidizing salts can materially affect sulfuric acid behavior and that chloride-contaminated submerged service may require higher-molybdenum alloys such as C-276, 622, 625, or 686. Haynes makes the same broader point: “real world” sulfuric acid service often departs from pure-acid lab behavior, so field validation is good engineering, not overkill.
For procurement teams, this has a practical implication. Do not ask only for alloy name. Ask for acid concentration range, maximum and upset temperature, aeration state, ferric/cupric contamination, chloride level, flow velocity, and whether the part sees crevices or deposits. Without that, any claim about Inconel 718 corrosion resistance in sulfuric acid is incomplete.
Final engineering takeaway
My conclusion is straightforward. Inconel 718 corrosion resistance in sulfuric acid is not poor by definition, but 718 is usually not the alloy I would lead with when sulfuric acid is the primary corrosion challenge. I would treat it as a strength-driven engineering compromise: acceptable in selected sulfuric environments, sometimes very useful, but not the default answer for hot or chemically complex acid service. If sulfuric acid is central to the duty, alloys such as 825, G-3, 625, C-276, or 686 usually deserve first review because their chemistry and published positioning align more directly with sulfuric acid performance.
At 28Nickel, the fastest way to make this decision correctly is to review the full service envelope, not just the alloy grade. If you are evaluating a shaft, fastener, tubing item, or vessel internal, send the sulfuric acid concentration, operating and upset temperature, impurity profile, required strength level, and fabrication route. That is enough to screen whether 718 is defensible or whether a sulfuric-acid-oriented alloy will give you a safer corrosion margin.
Domande e risposte correlate
1. Is Inconel 718 suitable for hot sulfuric acid service?
Usually not as a first choice when sulfuric acid is the main corrosive medium. Published sulfuric-acid guidance from nickel-alloy producers more often points engineers toward 825, G-3, C-276, 625, or 686, especially as temperature and concentration increase.
2. Why is 825 often preferred over 718 in sulfuric acid?
Because 825 is specifically positioned for sulfuric acid service and includes alloying additions, including molybdenum and copper, that are advantageous in sulfuric acid environments. Special Metals even provides sulfuric-acid iso-corrosion guidance for 825.
3. What data should I gather before selecting 718 for sulfuric acid duty?
At minimum: acid concentration, normal and upset temperature, oxidizing contaminants, chlorides, flow condition, crevice risk, corrosion allowance, and minimum mechanical property requirement. Those variables control whether 718 is a workable compromise or the wrong alloy entirely.