Chemical processing environments demand uncompromising material selection. When handling aggressive media like hot sulfuric, hydrochloric, or phosphoric acids, standard austenitic stainless steels undergo rapid anodic dissolution. The presence of halide ions aggressively breaches passive oxide layers, leading to catastrophic localized pitting. This is why specifying the correct nickel alloy for acidic environment operations is not just about extending equipment lifespan; it is a baseline for structural safety. Material failure in low pH systems often stems from a fundamental misunderstanding of how specific microstructural alloying elements interact with the thermodynamics of the corrosive media.

The metallurgical defense against acidic attack relies heavily on the synergistic alloying of Nickel (Ni), Molybdenum (Mo), and Chromium (Cr). High nickel baseline content provides the thermodynamic stability required to resist chloride stress corrosion cracking (CSCC), a pervasive threat in hot acid streams. When evaluating any nickel alloy for acidic environment services, engineers frequently reference the Pitting Resistance Equivalent Number (PREN). However, PREN alone is fundamentally insufficient when dealing with strictly reducing acids. Molybdenum shifts the corrosion potential in the noble direction, actively decelerating the anodic kinetics in non-oxidizing acids. Furthermore, the thermal history of the material dictates its field performance. Improper heat treatment can lead to the precipitation of deleterious topologically close-packed (TCP) phases, such as the Mu phase, along grain boundaries. This depletes adjacent regions of Mo and Cr, initiating severe intergranular corrosion (IGC).
| Grado di lega | Designazione UNS | Elementi chiave della lega | Corrosion Rate in Boiling 10% H2SO4 (mpy) | Primary Acid Application Focus |
| Lega 400 | N04400 | 63% Ni, 28-34% Cu | < 5.0 (Deaerated) | Hydrofluoric acid, Deaerated sulfuric |
| Lega 825 | N08825 | 38-46% Ni, 19-23% Cr, 2.5-3.5% Mo | < 10.0 | Phosphoric acid, Mixed acid streams |
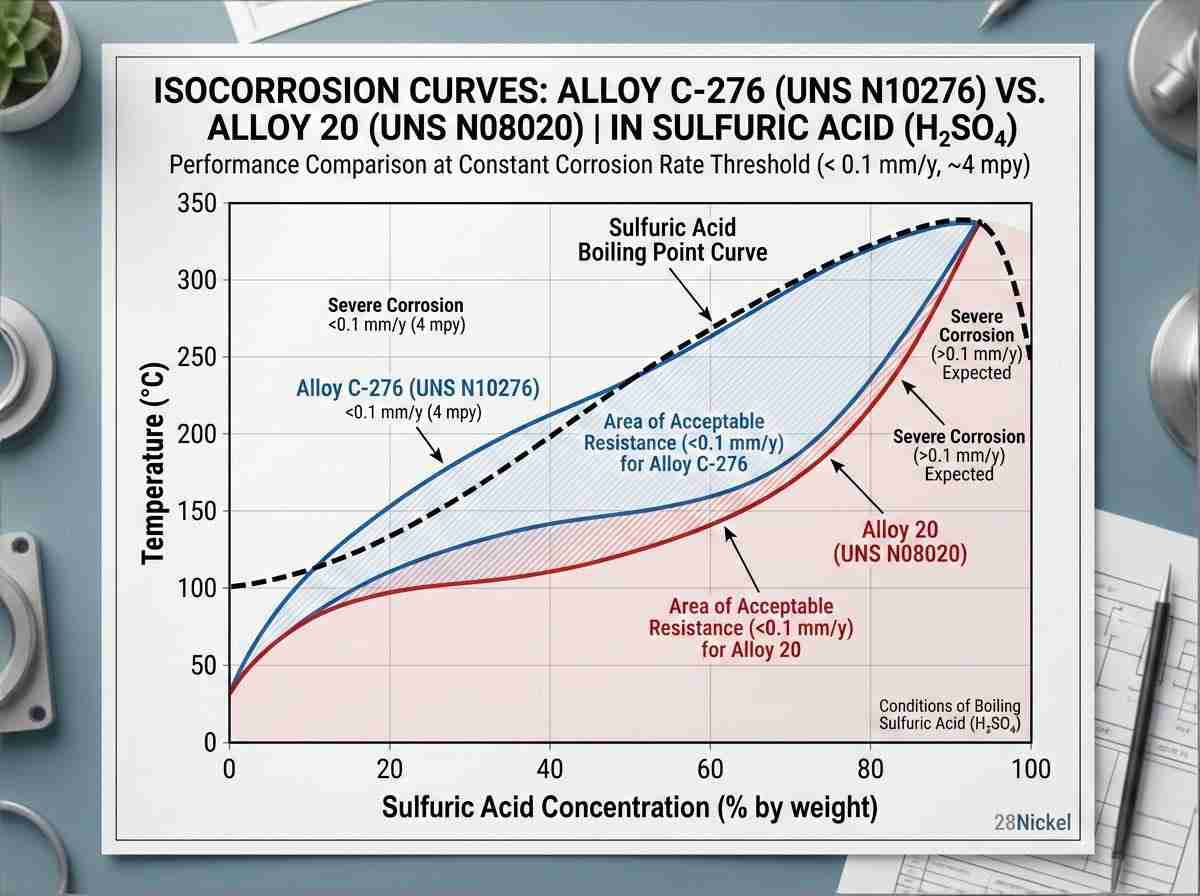
| Lega C-276 | N10276 | 57% Ni, 15-17% Mo, 14.5-16.5% Cr | < 2.0 | Contaminated hydrochloric & sulfuric acids |
Performance Metrics: The Ideal Nickel Alloy for Acidic Environment
Examining raw corrosion data reveals clear performance boundaries. In boiling 10% sulfuric acid, Alloy C-276 maintains a corrosion rate of less than 2 mpy. This exceptional resistance is primarily due to its high Molybdenum and 4% Tungsten (W) content. Tungsten acts synergistically with Mo to stabilize the passive film and inhibit localized attack under highly acidic conditions. Conversely, Alloy 400, a Ni-Cu system, excels in reducing hydrofluoric acid but suffers rapid, severe degradation in highly aerated, oxidizing acids. The introduced oxygen acts as an alternate cathodic reactant, severely driving the anodic dissolution.
Choosing a modern nickel alloy for acidic environment applications, like Alloy 22 (UNS N06022), mitigates the risk of intergranular attack through optimized composition. Alloy 22 can withstand complex, mixed-acid streams containing oxidizing impurities like ferric or cupric ions, which typically destroy pure Mo-bearing alloys like Alloy B-2. Additionally, mechanical integrity must be considered alongside chemical resistance. Components subjected to high-velocity acid streams experience erosion-corrosion, stripping away passive films faster than they can reform. Here, the solid-solution strengthening provided by Molybdenum not only increases yield strength but enhances the shear resistance of the protective oxide layer. Matching the specific nickel alloy for acidic environment conditions requires mapping the precise acid concentration, temperature profile, flow velocity, and aeration status directly onto an iso-corrosion chart.

Material specification in extreme pH conditions leaves zero margin for error. A nominal deviation in operating temperature or the unintended introduction of a trace catalyst can abruptly shift a material from the passive to the active state on the Pourbaix diagram. At 28Nickel, our metallurgical engineering team analyzes your specific process parameters—from fluid velocity to trace contaminants—to recommend the exact nickel alloy for acidic environment operation that ensures absolute structural integrity and process safety. Reach out to our technical support engineering team to review detailed iso-corrosion curves, stress mitigation strategies, and mechanical data tailored specifically to your reactor design parameters.
Domande e risposte correlate
Q1: Does a higher PREN guarantee better performance for a nickel alloy for acidic environment?
Not necessarily. The PREN formula (PREN=%Cr+3.3×%Mo+16×%N) primarily predicts resistance to localized pitting in chloride-bearing environments. In purely reducing acids, the base Nickel content and pure Molybdenum concentration dictate the corrosion rate far more than Chromium. Chromium is heavily relied upon only if the acid stream is oxidizing.
Q2: How does Titanium stabilization affect Alloy 825 in hot acid applications?
Alloy 825 utilizes Titanium additions to stabilize the microstructure against sensitization. The Titanium binds preferentially with carbon, preventing the formation of chromium carbides at the grain boundaries during high-temperature thermal cycles (like welding). This stabilization prevents intergranular attack when the alloy is subsequently exposed to aggressive acidic media.
Q3: What dictates the failure of Ni-Mo alloys (like Alloy B-2) in seemingly reducing acidic environments?
Alloy B-2 exhibits outstanding resistance to pure hydrochloric acid. However, it is extremely sensitive to oxidizing contaminants. Even trace amounts of ferric ions (Fe3+) or cupric ions (Cu2+) acting as oxidizing agents will catastrophically accelerate the corrosion rate of this specific nickel alloy for acidic environment services, shifting the electrochemical potential straight into the active dissolution range.