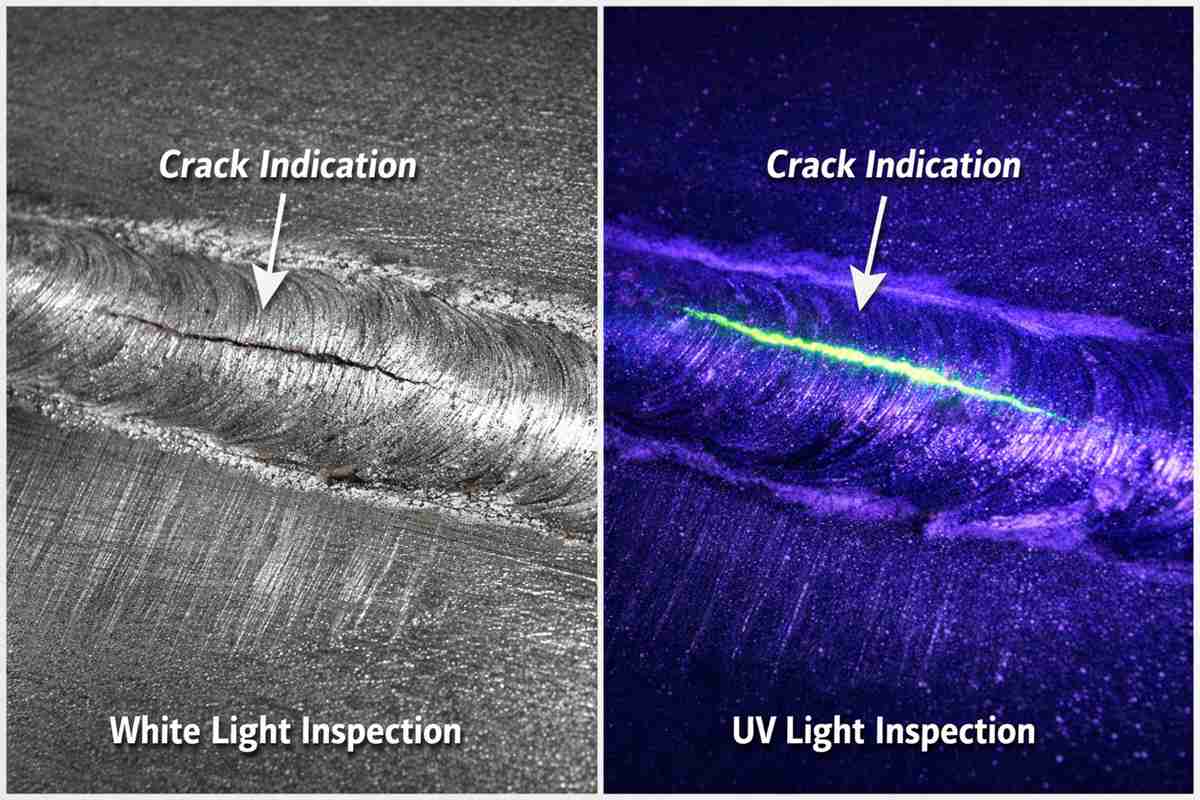
In the severe environments of modern chemical processing, material degradation is an ever-present threat to operational stability. Halides, localized high-temperature zones, and mixed acid streams relentlessly attack structural integrity, making standard austenitic stainless steels entirely obsolete. For engineers tasked with specifying materials for aggressive liquid or gaseous media, partnering with a knowledgeable ニッケル合金サプライヤー for chemical industry is not merely a standard supply chain decision; it is a critical metallurgical safeguard.
The true cost of a chemical reactor is not the initial capital expenditure, but the unplanned downtime caused by premature vessel failure. Pitting, crevice corrosion, and chloride-induced stress corrosion cracking (CISCC) dictate the lifespan of heat exchangers, scrubbers, and piping systems. Resolving these challenges requires analyzing them strictly through the lens of applied materials science.

Mitigating Localized Attack: Phase Stability
When evaluating materials for wet chlorine gas or hot concentrated sulfuric acid, phase stability and precise elemental composition are paramount. High nickel content fundamentally stabilizes the austenitic matrix, which drastically reduces the susceptibility to chloride-ion stress corrosion cracking. However, localized attack requires specific alloying additions to halt pitting mechanisms. Molybdenum and tungsten fundamentally improve the Pitting Resistance Equivalent Number (PREN). A competent nickel alloy supplier for chemical industry must provide detailed insights into how these elements interact during thermal cycling to prevent the precipitation of detrimental phases, such as the sigma or mu phases, which drastically embrittle the structural matrix.
Furthermore, sensitization in the heat-affected zone (HAZ) during welding remains a primary failure mode in chemical plant fabrication. If carbon and silicon are not strictly controlled during the melt phase, grain boundary carbide precipitation occurs. The right nickel alloy supplier for chemical industry understands that specific low-carbon grades (like Alloy C-276 or Alloy 59) must be rigorously tested for intergranular corrosion resistance per stringent ASTM G28 specifications.
| 合金グレード | UNS指定 | ニッケル(%) | Cr (%) | モリブデン (%) | PREN(約) | Primary Chemical Resistance Profile |
| 合金C-276 | N10276 | バル. | 15.5 | 16.0 | 68 | Exceptional in wet chlorine, hypochlorites, and mixed acids. |
| アロイ22 | N06022 | バル. | 21.0 | 13.5 | 65 | Superior resistance to oxidizing aqueous media and pitting. |
| アロイ625 | N06625 | バル. | 21.5 | 9.0 | 51 | High fatigue strength; resists chloride-ion stress corrosion. |
| アロイ400 | N04400 | バル. | – | – | 該当なし | Excellent in hydrofluoric acid and harsh halide environments. |
High-Temperature Mechanical Properties
Chemical synthesis often demands high-pressure reactions at elevated temperatures. Solid solution strengthened alloys provide excellent creep and rupture strength up to 1200°F (650°C). Selecting the correct metallurgical profile requires cross-referencing isocorrosion charts with the ASME Boiler and Pressure Vessel Code (BPVC) maximum allowable stress values. A specialized nickel alloy supplier for chemical industry will never guess; they will utilize empirical corrosion data and mechanical testing reports to validate the material’s viability under your specific operational stress loads.
Reducing environments, such as hydrochloric and hydrofluoric acids, necessitate copper-nickel or pure nickel solid solutions. Conversely, oxidizing environments demand higher chromium content to maintain the passive oxide layer. Navigating these conflicting requirements is where a true nickel alloy supplier for chemical industry demonstrates tangible engineering value. For instance, the presence of even trace amounts of ferric or cupric ions can drastically shift a highly reducing acidic environment into an oxidizing one, accelerating the corrosion rate of specific alloys. Anticipating these dynamic process changes requires deep material science expertise.
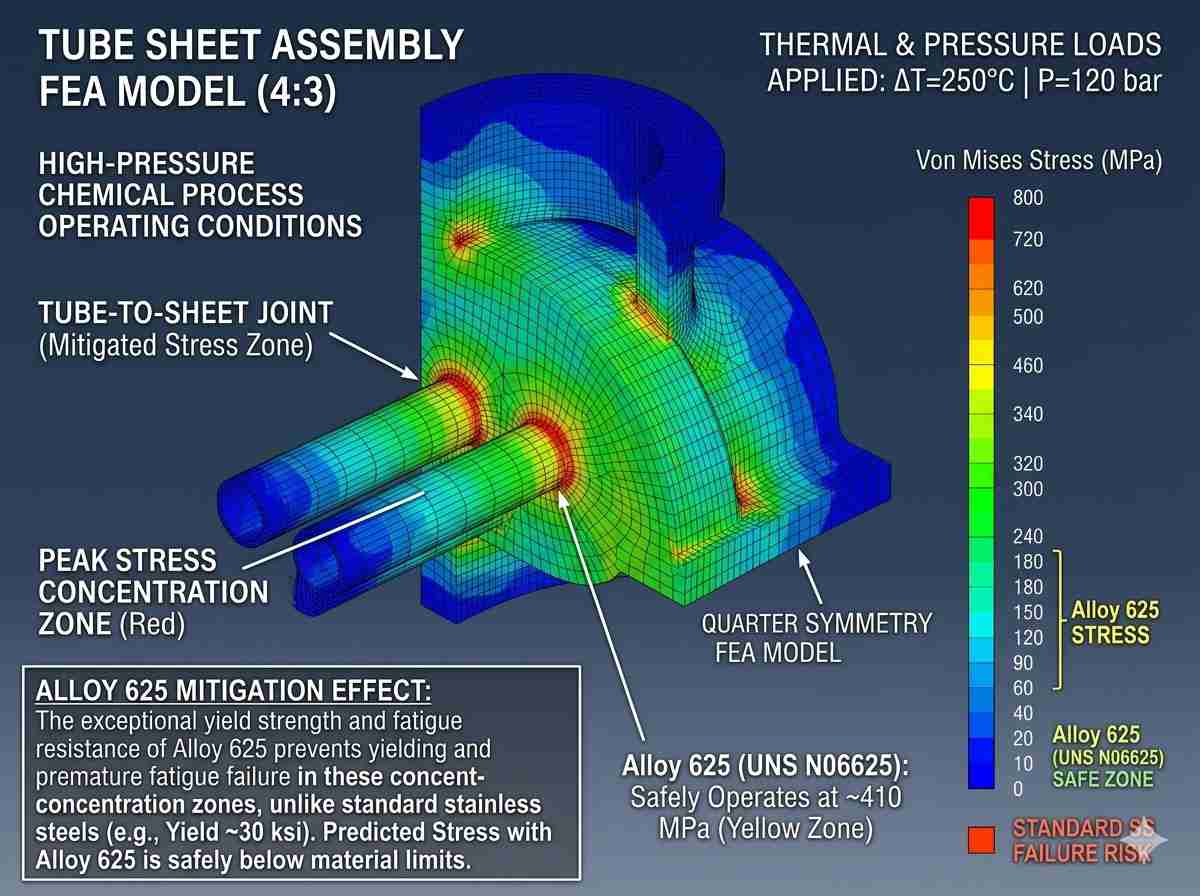
Engineering Reliability Through Precise Specification
Achieving a high Mean Time Between Failures (MTBF) in chemical processing relies entirely on microstructural integrity. Material selection is an exact science, heavily reliant on accurate chemical composition control and precise solution annealing processes. Choosing a technically proficient nickel alloy supplier for chemical industry ensures that every plate, tube, and forging meets the rigorous metallurgical demands of your specific flow conditions.
The consequences of material failure in chemical processing are catastrophic. At 28Nickel, we operate strictly as engineers serving engineers. We provide the hard data, the microstructural analysis, and the deep metallurgical knowledge required to secure your processing equipment against severe corrosion. If your facility is struggling with premature equipment degradation, partnering with an expert nickel alloy supplier for chemical industry is your most effective countermeasure. When you engage with an experienced nickel alloy supplier for chemical industry, you gain a partner in risk mitigation. Reach out to our engineering team to discuss your specific corrosive environment and let us collaborate on a resilient material solution.
関連Q&A
Q: How does the PREN value influence material selection for chloride-rich chemical environments?
A: The Pitting Resistance Equivalent Number (PREN) mathematically calculates localized corrosion resistance based on Chromium, Molybdenum, and Nitrogen levels. In chloride-rich chemical media, an alloy with a PREN > 40 is typically required to maintain electrochemical passivity and resist localized pitting attack.
Q: Why is carbon content strictly controlled in ニッケル合金 used for hot sulfuric acid applications?
A: High carbon content leads to carbide precipitation at the grain boundaries during welding, a process known as sensitization. This creates chromium-depleted zones, making the heat-affected zone (HAZ) highly susceptible to intergranular corrosion in aggressive hot acids. Low-carbon grades prevent this microstructural degradation.
Q: What is the primary metallurgical advantage of Alloy 22 over C-276 in oxidizing media?
A: While both are exceptional materials, Alloy 22 contains significantly higher chromium (~21% compared to ~15.5%). This enhanced chromium content drastically improves its ability to form and maintain a protective passive oxide film in highly oxidizing environments, such as mixed acids containing nitric acid or dissolved ferric ions.