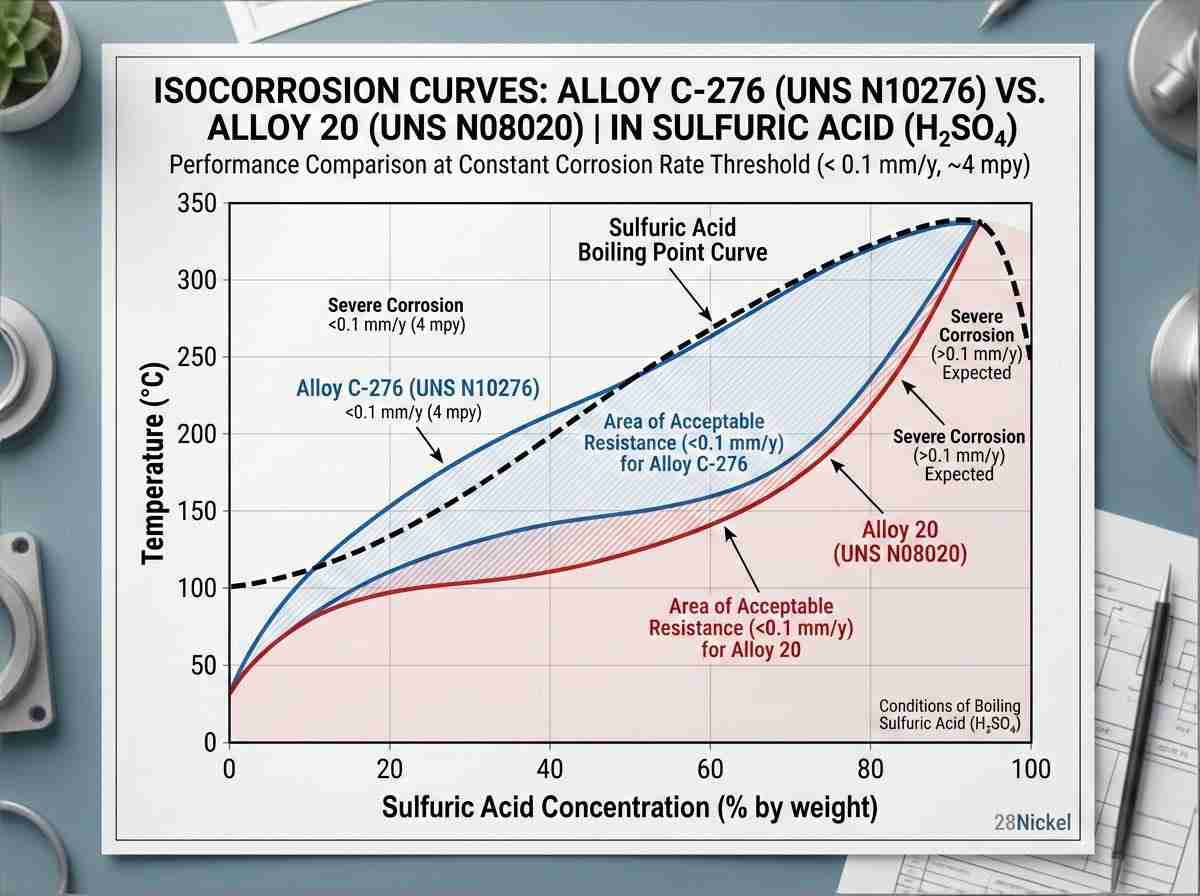
As fábricas de produtos químicos operam em ambientes excecionalmente agressivos. A corrosão por picadas, a corrosão em fendas e a fissuração por corrosão sob tensão (SCC) são ameaças constantes ao funcionamento contínuo. Quando se lida com meios agressivos, como ácido sulfúrico a ferver ou soluções de cloreto a quente, os aços inoxidáveis austeníticos padrão simplesmente falham. É exatamente aqui que a seleção precisa da liga de níquel para o processamento químico se torna um requisito crítico de engenharia. A escolha certa determina o ciclo de vida de um vaso de reator. A presença de iões halogenados complica muito a especificação dos materiais. É frequente vermos ataques localizados que se iniciam nos limites dos grãos ou por baixo de depósitos. A seleção eficaz de ligas de níquel para processamento químico nestes cenários depende do Número Equivalente de Resistência a Pitting (PREN). As ligas ricas em molibdénio e tungsténio, tais como Hastelloy C-276 (UNS N10276), proporcionam uma resistência excecional. O molibdénio estabiliza a película passiva contra a penetração de iões de cloreto. Por outro lado, se o ambiente for puramente redutor, como o cloreto de hidrogénio seco, a liga 400 (UNS N04400), aproveitando o seu elevado teor de cobre, demonstra uma estabilidade termodinâmica superior. A variação dos mecanismos de degradação significa que a seleção de ligas de níquel para processamento químico não pode ser feita através de fichas de dados genéricas. Requer uma análise rigorosa das espécies iónicas específicas, gradientes de temperatura e velocidades de fluxo no fluxo do processo.
Variações metalúrgicas na seleção de ligas
Vejamos a química fundamental que dita estas escolhas. Um elevado teor de níquel suprime diretamente a fissuração por corrosão sob tensão por cloreto, uma vulnerabilidade conhecida dos aços inoxidáveis da série 300. Ao ajustar o crómio, ganhamos resistência a agentes oxidantes como o ácido nítrico. A estabilidade da matriz é fundamental. A seleção incorrecta de ligas de níquel para processamento químico resulta frequentemente do facto de se ignorarem os vestígios de contaminantes. Uma quantidade vestigial de iões férricos num fluxo de ácido clorídrico pode mudar instantaneamente o ambiente de redutor para oxidante, tornando subitamente vulnerável uma liga pesada de molibdénio previamente especificada.
| Grau da liga | Designação UNS | Ni (%) | Cr (%) | Mo (%) | PREN (Aprox.) | Aplicação ambiental primária |
| Liga C-276 | N10276 | 57.0 | 16.0 | 16.0 | > 45 | Ácidos mistos graves, cloretos contaminados |
| Liga 625 | N06625 | 58.0 | 21.0 | 9.0 | ~ 40 | Ambientes de corrosão por picadas e fendas |
| Liga 825 | N08825 | 42.0 | 21.5 | 3.0 | ~ 31 | Manuseamento de ácido sulfúrico e fosfórico |
| Liga 400 | N04400 | 63.0 | – | – | N/A | Ácido fluorídrico e salmoura desaerada |
Modos de degradação a alta temperatura
Para além da corrosão aquosa, as reacções de gás a alta temperatura apresentam outra camada de complexidade. A oxidação, a sulfidação e a carburação degradam rapidamente a integridade mecânica. Na engenharia de componentes internos para fornos de pirólise ou regeneradores catalíticos, a seleção de ligas de níquel para processamento químico muda o foco para formadores de carbonetos e estabilização de incrustações de alumina ou crómio. A liga 600 (UNS N06600), por exemplo, tem um desempenho admirável em cloro quente e seco até 540°C, mas apresenta riscos de sulfidação se houver espécies de enxofre presentes. Nestes ambientes de gás misto, o equilíbrio preciso de crómio e alumínio torna-se o fator decisivo.
O impacto do fabrico
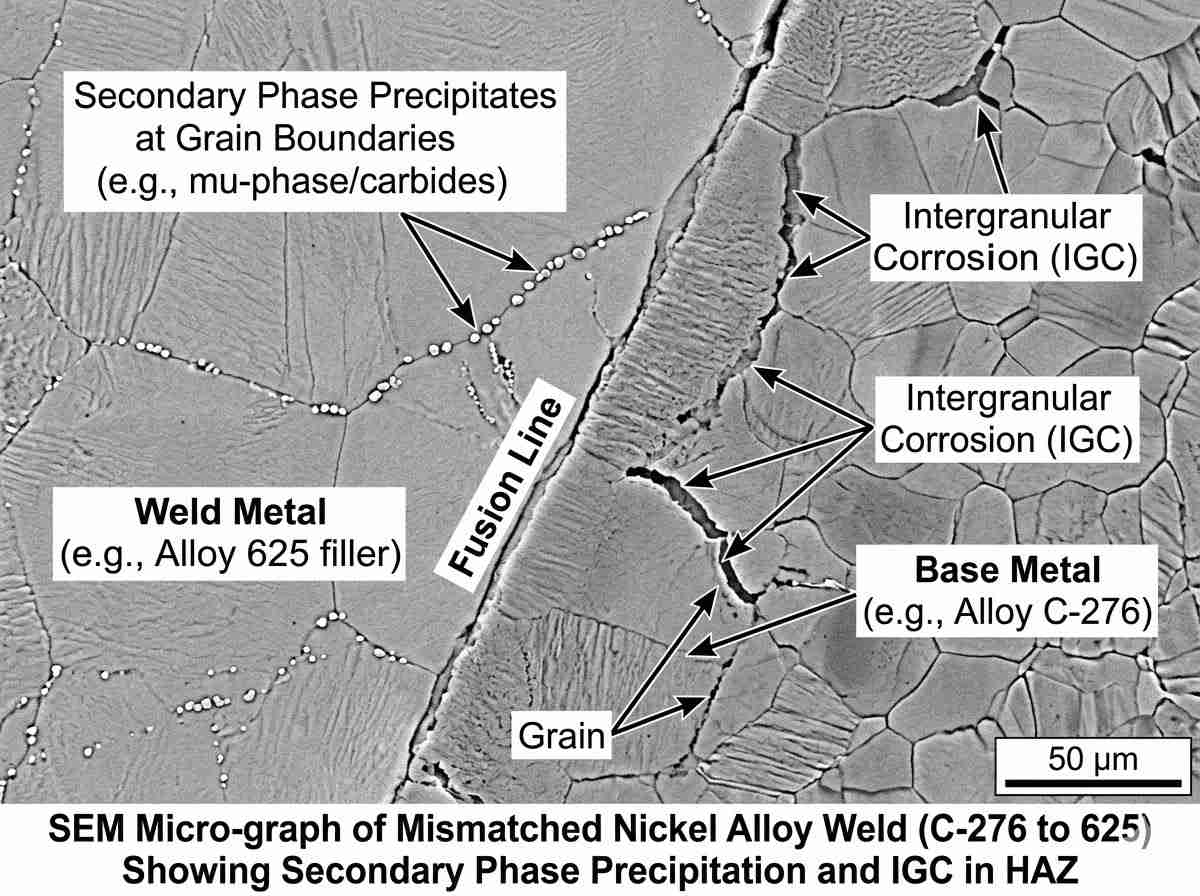
Mesmo a mais rigorosa seleção de ligas de níquel para a estratégia de processamento químico pode ser totalmente desfeita por técnicas de fabrico inadequadas. A soldadura introduz gradientes térmicos significativos, criando uma Zona Afetada pelo Calor (ZAC) onde pode ocorrer a precipitação de fases secundárias. Por exemplo, a precipitação da fase mu ou de carbonetos prejudiciais nos limites dos grãos reduz drasticamente a resistência à corrosão localizada. Por conseguinte, a especificação do metal de adição correto - frequentemente ligado em excesso em comparação com o material de base - é tão crítica como a especificação primária. Defendemos vivamente as entradas de calor controladas e, quando necessário, o tratamento térmico pós-soldadura (PWHT) para restaurar a homogeneidade microestrutural. Os engenheiros devem integrar as restrições de fabrico na fase inicial da seleção da liga de níquel para a matriz de processamento químico, a fim de evitar vulnerabilidades na zona de fusão.
Otimizar a fiabilidade através da engenharia
Como engenheiros de materiais, temos de nos basear em dados empíricos, testes rigorosos e modelação exacta de processos. A penalização económica de uma especificação mal calculada é catastrófica, resultando em interrupções não planeadas e riscos de segurança críticos. É imperativo modelar a termodinâmica do fluido exato do processo. Em última análise, a seleção ideal de ligas de níquel para o processamento químico não se trata apenas de encontrar o material mais resistente à corrosão; trata-se de encontrar o perfil metalúrgico preciso que garanta uma fiabilidade previsível sem um excesso de engenharia. Na 28Nickel, a nossa equipa de engenharia de materiais analisa profundamente os seus parâmetros operacionais específicos. Fornecemos a validação técnica necessária para proteger a sua infraestrutura. Contacte o nosso departamento de engenharia para discutir os seus desafios exactos de degradação, analisar os seus dados de isocorrosão e garantir o apoio técnico direcionado que o seu projeto exige.
Perguntas e respostas relacionadas:
Q1: Como é que a contaminação por vestígios de flúor afecta a seleção de ligas de níquel para processamento químico?
A: Os fluoretos atacam agressivamente a camada passiva de óxido de muitas ligas que contêm crómio. Nestes casos, são necessárias ligas com elevado teor de cobre, como a liga 400, ou variações extremamente elevadas de molibdénio, em função da presença simultânea de espécies oxidantes.
Q2: Porque é que o valor PREN, por si só, não é suficiente para a seleção de ligas de níquel para processamento químico?
A: O PREN prevê apenas a resistência à corrosão localizada em ambientes ricos em cloretos a temperaturas ambiente ou moderadamente elevadas. Não tem em conta as taxas gerais de corrosão em ácidos redutores, os riscos de fissuração por corrosão sob tensão ou a sulfidação a alta temperatura.
Q3: Qual é o limiar crítico de níquel necessário para evitar a fissuração por corrosão sob tensão por cloreto?
A: Os dados empíricos sugerem que as ligas com um teor de níquel superior a 42% (como a liga 825) apresentam uma imunidade significativa à SCC induzida por cloreto, enquanto as ligas com um teor de níquel próximo de 60% (como a liga 625) oferecem uma imunidade virtual na maioria dos fluxos de processos químicos normais.



