In the demanding world of industrial engineering, nickel alloy corrosion resistance is the primary reason these materials are selected for the most punishing environments on Earth. From the depths of the ocean to the high-heat zones of chemical processing plants, nickel alloys stand as a sentinel against material degradation. Unlike standard steels, these alloys are designed to maintain their structural integrity where others succumb to oxidation, pitting, and stress-related failure. At 28Nickel, we understand that selecting the right material isn’t just about cost; it is about the long-term reliability of your critical infrastructure.
The Science Behind Nickel Alloy Corrosion Resistance
The superior performance of nickel alloy corrosion resistance stems from the unique atomic structure of nickel itself. Nickel possesses a face-centered cubic (FCC) lattice, which allows it to remain ductile and tough across a massive temperature range. More importantly, nickel has a natural ability to form a passive, protective oxide layer on its surface.
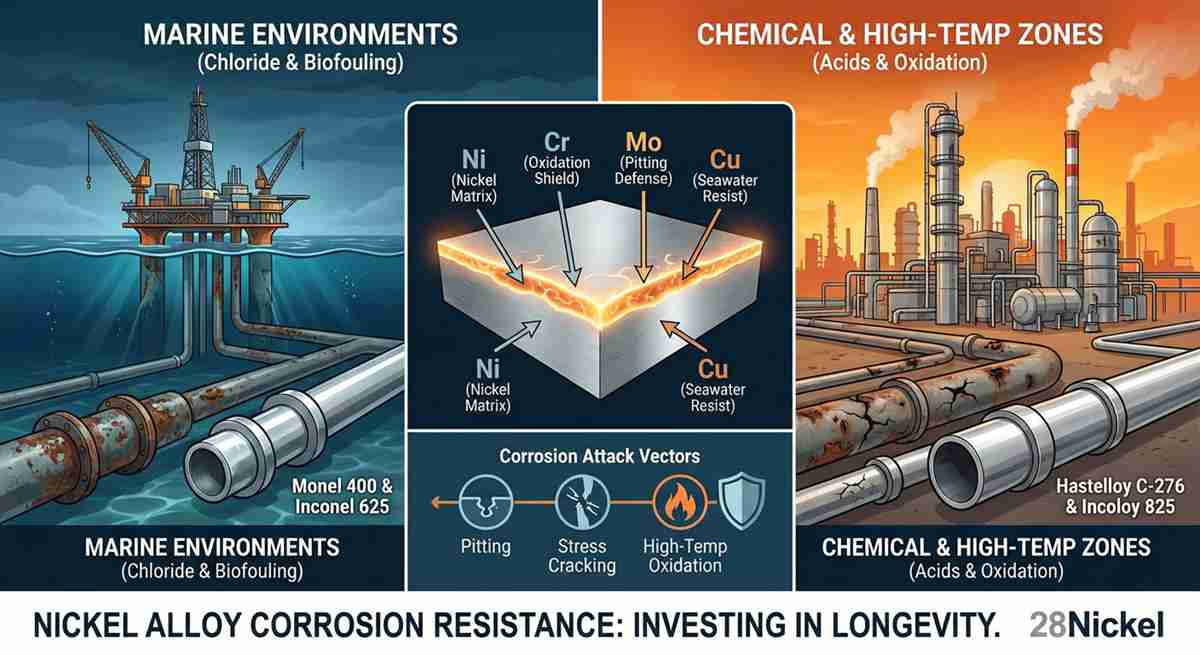
However, “pure” nickel is only the beginning. To enhance nickel alloy corrosion resistance for specific industrial challenges, elements like Chromium (Cr), Molybdenum (Mo), and Copper (Cu) are added.
-
Chromium is essential for resistance in oxidizing environments (like nitric acid).
-
Molybdenum is the secret weapon against localized attacks like pitting and crevice corrosion in chloride-rich environments.
-
Copper is added to alloys like Monel 400 to provide unmatched resistance to seawater and hydrofluoric acid.
Types of Attacks Handled by Nickel Alloy Corrosion Resistance
Not all corrosion is created equal. Understanding how nickel alloy corrosion resistance combats different “attack vectors” is crucial for material selection.
-
Pitting and Crevice Corrosion: In stagnant fluids containing halides (like salt water), standard stainless steel often develops small, deep holes (pits). High-performance nickel alloys with high PREN (Pitting Resistance Equivalent Number) scores utilize molybdenum and tungsten to keep these surfaces pristine.
-
Stress Corrosion Cracking (SCC): This is the “silent killer” of many alloys. Under specific stress and corrosive conditions, a metal can fail suddenly without warning. Nickel’s high nickel content (usually above 30%) provides an almost complete immunity to chloride-induced SCC.
-
High-Temperature Oxidation: Many industrial processes operate at temperatures where metals literally “burn” in air. The nickel alloy corrosion resistance at high temperatures relies on the formation of a stable, adherent scale of chromium oxide or aluminum oxide that prevents further oxygen penetration.
Comparison Table: Nickel Alloy Corrosion Resistance by Grade
| Alloy Grade | Primary Environments | Key Benefit | Resistance to Pitting |
| Monel 400 | Seawater, Hydrofluoric Acid | Excellent in reducing media | Moderate |
| Inconel 625 | Marine, Aerospace, Chemical | High strength + Oxidation resistance | Excellent |
| Hastelloy C-276 | Harsh Chemicals, Flue Gas | Versatile in “dirty” environments | Superior |
| Incoloy 825 | Oil & Gas, Acid Production | Resistance to sulfuric/phosphoric acids | High |
Environmental Factors Influencing Nickel Alloy Corrosion Resistance
It is a common misconception that one alloy fits all. The effectiveness of nickel alloy corrosion resistance is heavily dependent on the “chemistry of the soup.” For instance, an alloy that performs beautifully in a reducing acid (like hydrochloric) might fail rapidly in an oxidizing acid (like nitric) if it lacks sufficient chromium.
Temperature is another critical variable. As temperature increases, the rate of chemical reactions accelerates. Superior nickel alloy corrosion resistance ensures that the protective oxide film remains stable even as thermal energy tries to disrupt the molecular bonds of the metal surface. This is why 28Nickel focuses on sourcing materials that meet rigorous international standards (ASTM/ASME), ensuring that the chemical composition is precise enough to guarantee these properties.
Conclusion: Investing in Longevity
Choosing a material based on nickel alloy corrosion resistance is an investment in safety and uptime. While the initial cost of nickel-based materials is higher than carbon steel, the “total cost of ownership” is significantly lower due to reduced maintenance, fewer replacements, and the prevention of catastrophic environmental leaks. At 28Nickel, we take pride in providing the technical expertise to help you navigate these complex metallurgical choices.
Related Q&A
Q1: Why is nickel better than stainless steel for corrosion?
While stainless steel is effective, nickel alloy corrosion resistance is superior in extreme temperatures and highly acidic or chloride-rich environments where stainless steel suffers from pitting or stress cracking.
Q2: Does “corrosion resistant” mean the alloy will never rust?
No metal is 100% “corrosion-proof.” However, the high nickel alloy corrosion resistance means the degradation rate is so slow that the material remains functional for decades in environments that would destroy other metals in weeks.
Q3: Which nickel alloy is best for marine environments?
Monel 400 and Inconel 625 are industry favorites for marine applications due to their exceptional nickel alloy corrosion resistance against salt water and biofouling.



