Designing pressure vessels and piping systems for aggressively corrosive environments leaves absolutely zero room for metallurgical guesswork. When engineers face pitting, stress corrosion cracking (SCC), or extreme thermal oxidation, the material selection matrix inevitably points toward nickel-based superalloys. Specifically, the ongoing debate of inconel vs hastelloy vs monel dictates the lifecycle of critical components in chemical processing plants, aerospace turbine engines, and offshore oil platforms. Understanding the specific atomic lattice structures, elemental synergies, and failure thresholds of these distinct alloy families is non-negotiable for any serious materials engineer.
Selecting the correct grade is not about finding the inherently “strongest” metal; it is about matching the precise thermodynamic stability of the alloy to the chemical kinematics of your operating environment. To effectively resolve the inconel vs hastelloy vs monel dilemma, we must break down their fundamental alloying principles.

Inconel vs Hastelloy vs Monel: Metallurgical Foundations
Let us dissect the alloying logic at the elemental level. When evaluating inconel vs hastelloy vs monel, you are essentially comparing the long-term performance of Ni-Cr, Ni-Mo/Ni-Cr-Mo, and Ni-Cu metallic systems under extreme kinetic stress.
Inconel (such as Alloy 625 or 718) relies heavily on its high chromium content to form a robust, self-repairing passive oxide layer. This mechanism makes the Inconel family exceptional in severe oxidizing environments. Furthermore, the deliberate addition of molybdenum and niobium provides significant solid-solution stiffening of the face-centered cubic (FCC) nickel-chromium matrix. In age-hardenable grades like 718, the precipitation of gamma double prime (γ′′) phases yields phenomenal tensile strength and structural integrity at elevated temperatures.
Hastelloy (including highly specified grades like C-276, B-2, and C-22) pushes molybdenum and tungsten mass fractions much higher. This is a highly calculated metallurgical choice engineered specifically to combat aggressive localized corrosion attacks—namely pitting and crevice corrosion. Hastelloy excels in severe reducing acids, such as wet chlorine gas and boiling hydrochloric environments, where other austenitic alloys rapidly undergo active dissolution.
Monel (like Alloy 400 and K-500), a primarily nickel-copper solid solution alloy, operates on entirely different thermodynamic principles. It lacks the chromium additions of its counterparts but exhibits extraordinary resistance to hydrofluoric acid and severe marine fouling. This performance is largely due to the inherent thermodynamic stability of the Ni-Cu metallic bond. To truly quantify the inconel vs hastelloy vs monel performance gap, we must examine the empirical mechanical data.
| Alloy Family | Primary Alloying Elements | Typical PREN | Yield Strength (MPa, Min) | Ultimate Tensile Strength (MPa, Min) | Primary Survival Environment |
| Inconel (625) | Ni, Cr (20%), Mo, Nb | ~50 | 414 | 827 | Severe oxidizing acids, high-temp oxidation up to 980°C |
| Hastelloy (C-276) | Ni, Mo (15%), Cr, W | ~68 | 283 | 690 | Aggressive reducing acids, wet chlorine, severe chlorides |
| Monel (400) | Ni, Cu (30%), Fe | N/A (Low Cr) | 195 | 480 | Hydrofluoric acid, high-velocity seawater, fluorine gas |
Evaluating Inconel vs Hastelloy vs Monel in Active Media
Analyzing the rigorous mechanical and chemical data above, the inconel vs hastelloy vs monel comparison becomes a highly specific matter of matching the elemental phase profile to the corrosive fluid dynamics.
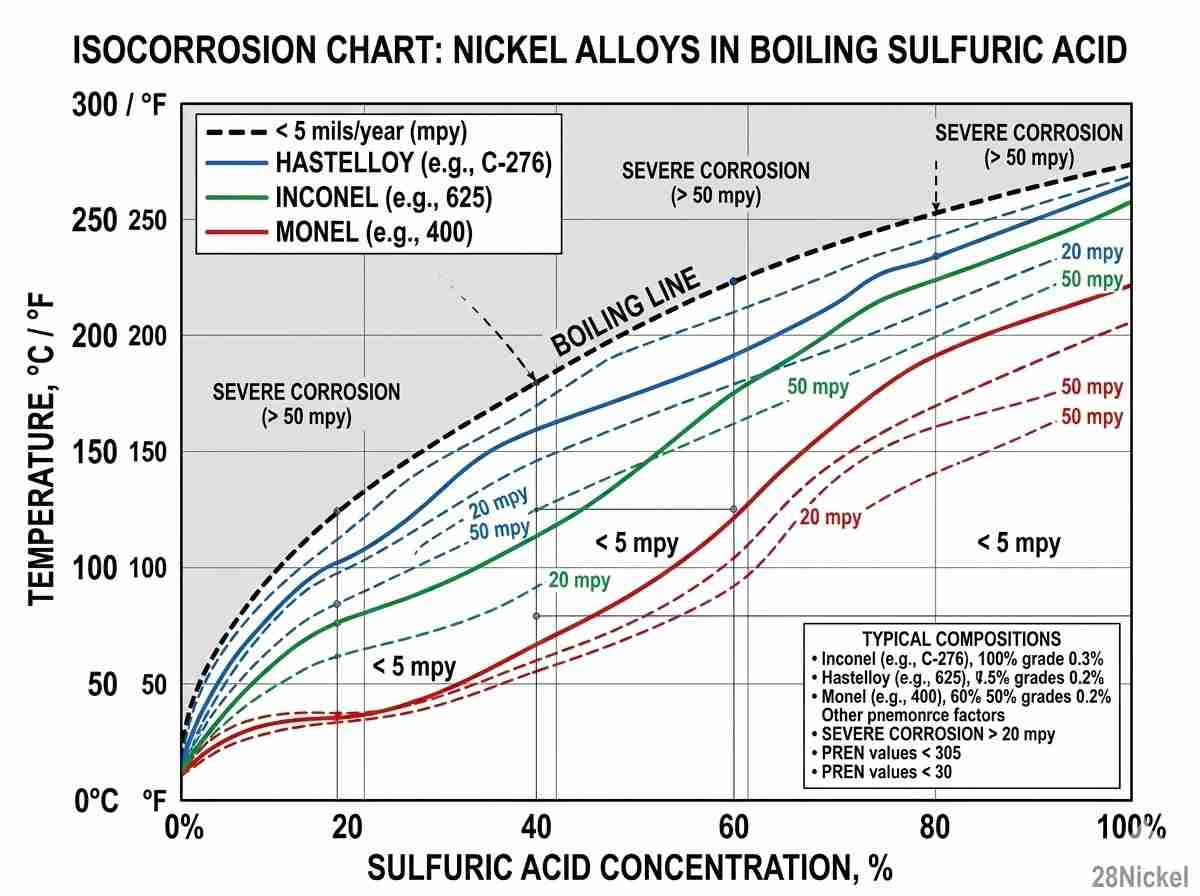
In hot, concentrated sulfuric acid applications, the isocorrosion curves clearly dictate the winner. Hastelloy C-276 maintains its passivity across a remarkably wide range of concentrations and thermal gradients, significantly outperforming Inconel in boiling states. The heavily enriched molybdenum matrix effectively halts chloride-induced stress corrosion cracking before intergranular micro-fissures can propagate.
However, if the operational environment introduces severe thermal cycling and high-temperature creep exceeding 980°C (1800°F), the paradigm shifts entirely. Inconel’s chromium-oxide layer remains tenaciously intact, effectively preventing high-temperature oxidation and internal carburization. In these specific aerodynamic or industrial exhaust applications, Hastelloy may face microstructural instability or high-temperature embrittlement over time, making Inconel the superior engineering specification.
Meanwhile, Monel dominates the highly specific niche of hydrofluoric acid alkylation units. Neither Inconel nor Hastelloy can economically match Monel 400’s exceptionally low corrosion rate in anhydrous HF. Under active reducing conditions, the high copper content in Monel provides a localized cathodic reaction that thermodynamically protects the base nickel matrix from degradation.
The Final Engineering Verdict
Therefore, the definitive answer to the inconel vs hastelloy vs monel engineering challenge lies exclusively in the exact acid concentration, temperature gradient, and halogen presence of your process stream. Miscalculating the Pitting Resistance Equivalent Number (PREN) or ignoring phase stability transitions at peak operating temperatures will inevitably lead to catastrophic localized failure. Advanced material specification requires rigorous failure analysis and empirical fluid data, not broad industry assumptions.
At 28Nickel, our senior materials engineering team possesses the deep metallurgical expertise required to rigorously analyze your technical drawings and chemical flow states. By mapping out the exact thermal and corrosive loads of your application, we can specify the exact superalloy grade required to ensure absolute structural integrity. Connect with our engineering desk today to share your operational parameters and discuss the technical material specifications for your next critical project.
Related Q&A
Q1: How does the PREN value influence the choice in an inconel vs hastelloy vs monel comparison for severe seawater applications? A: PREN (Pitting Resistance Equivalent Number) mathematically favors Hastelloy and Inconel due to their heavily alloyed Chromium and Molybdenum matrices. Hastelloy C-276 (PREN ~68) and Inconel 625 (PREN ~50) offer virtual immunity to localized pitting in stagnant marine environments. Monel cannot be evaluated via PREN due to its lack of chromium, but it relies on overall thermodynamic stability to resist high-velocity seawater, though it remains highly susceptible to localized pitting under biological fouling or stagnant conditions.
Q2: In high-temperature gas turbines, why is the inconel vs hastelloy vs monel debate usually resolved strictly in favor of Inconel? A: The primary failure mode in modern gas turbines is high-temperature creep combined with severe oxidation. Inconel grades (like 718) form a highly stable, self-adherent Cr2O3 scale that protects the internal matrix up to 980°C. Furthermore, Inconel’s controlled gamma prime (γ′) and gamma double prime (γ′′) precipitates provide exceptional intergranular creep-rupture strength. Hastelloy is metallurgically optimized for severe aqueous corrosion rather than extreme thermal oxidation, and Monel begins to lose structural integrity rapidly above 480°C.
Q3: Can Monel effectively replace Hastelloy in aggressively reducing acid environments? A: It depends strictly on the specific molecular makeup of the acid. The inconel vs hastelloy vs monel decision here requires pinpoint precision. Monel is vastly superior in hydrofluoric (HF) acid due to its copper content forming a dense, protective fluoride film. However, in boiling hydrochloric (HCl) or reducing sulfuric media heavily contaminated with highly active chlorides, Hastelloy (specifically B-type or C-276) is absolutely mandatory. Monel will suffer rapid uniform dissolution in highly aerated or oxidizing acid mixtures where Hastelloy remains fully passive.



