Selecting materials for turbine blades, chemical reactors, or aerospace exhaust systems is rarely a simple task. Engineers constantly battle thermal fatigue, creep deformation, and aggressive oxidation. Establishing the precise nickel alloy working temperature range is the absolute baseline for safe, continuous, and reliable operation. If you miscalculate this operational window, catastrophic grain boundary failure and severe yield strength degradation are almost inevitable.
To prevent these failures, understanding the nickel alloy working temperature range requires looking far beyond basic melting points. The actual operational limit of a superalloy is dictated by complex metallurgical mechanisms, specifically the stability of its microstructural phases under sustained thermal stress. We must evaluate solid solution strengthening versus precipitation hardening to determine the true upper limits.
Defining the Nickel Alloy Working Temperature Range
When specifying materials for extreme environments, metallurgists generally divide these alloys into two primary categories: solid-solution-strengthened alloys and precipitation-hardened (age-hardened) alloys. The difference in their strengthening mechanisms directly dictates their maximum operational thresholds.
Solid-solution alloys, such as Inconel 600 and Hastelloy X, rely on elements like chromium, molybdenum, and iron dissolving into the nickel matrix. Because they do not rely on microscopic precipitates for strength, their structural integrity remains stable at exceptionally high heat. Consequently, their nickel alloy working temperature range can safely extend up to 1150°C to 1200°C for applications requiring oxidation resistance rather than extreme high-temperature load-bearing capacity.
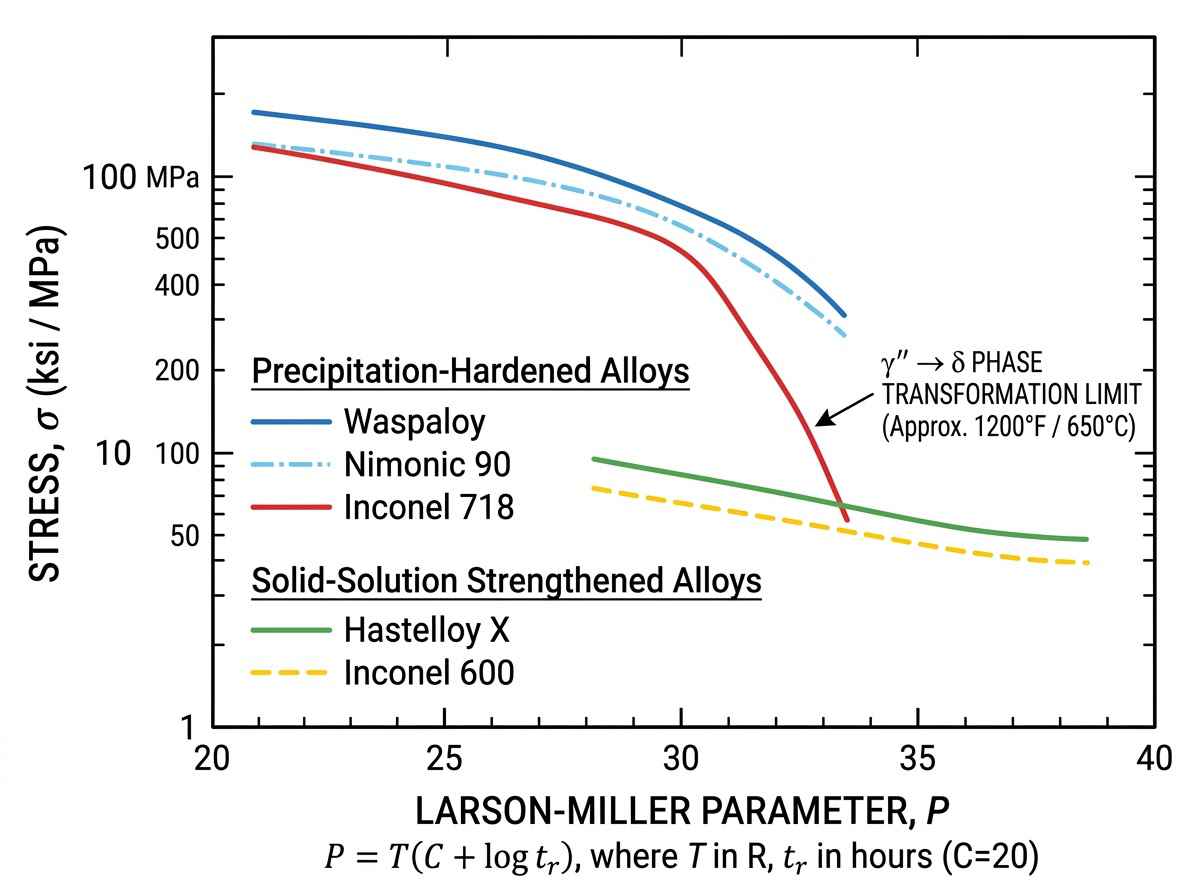
Conversely, precipitation-hardened alloys like Inconel 718 and Waspaloy achieve superior yield strength through the precipitation of γ′ (gamma prime, Ni3(Al,Ti)) and γ′′ (gamma double prime, Ni3Nb) phases. While these phases provide massive tensile strength at elevated temperatures, they are thermodynamically unstable above certain thresholds. If Inconel 718 exceeds 650°C for extended periods, the γ′′ phase begins to coarsen and transform into the stable, but weaker, δ (delta) phase. Therefore, the effective nickel alloy working temperature range for highly stressed precipitation-hardened components is narrower, strictly limited by phase transformation kinetics.
| Alloy Grade | Strengthening Mechanism | Key Alloying Elements | Safe Max Continuous Temp (°C) | Primary Degradation Mode at Limit |
| Inconel 600 | Solid Solution | Ni, Cr, Fe | 1095 | Oxidation / Carburization |
| Inconel 625 | Solid Solution (Mo, Nb) | Ni, Cr, Mo, Nb | 982 | Microstructural sensitization |
| Inconel 718 | Precipitation ($\gamma”$) | Ni, Cr, Fe, Nb, Mo | 650 | $\gamma”$ to $\delta$ phase transformation |
| Hastelloy X | Solid Solution | Ni, Cr, Fe, Mo | 1177 | Long-term embrittlement |
| Nimonic 90 | Precipitation ($\gamma’$) | Ni, Cr, Co, Ti, Al | 900 | $\gamma’$ coarsening / Creep |
Microstructural Stability and Creep Resistance
Beyond simple phase transformations, creep rupture strength is often the deciding factor in engineering applications. Creep—the time-dependent permanent deformation of a material under constant mechanical stress at high temperatures—accelerates exponentially as thermal loads increase.
Evaluating the nickel alloy working temperature range for a specific industrial application means analyzing the Larson-Miller parameter of the chosen material. Alloys with high cobalt, tungsten, and tantalum additions, such as certain cast superalloys, impede dislocation movement and grain boundary sliding, pushing the creep resistance threshold higher. Furthermore, the formation of Topologically Close-Packed (TCP) phases (such as σ or μ phases) during prolonged high-temperature exposure must be avoided, as these brittle phases act as crack initiation sites.
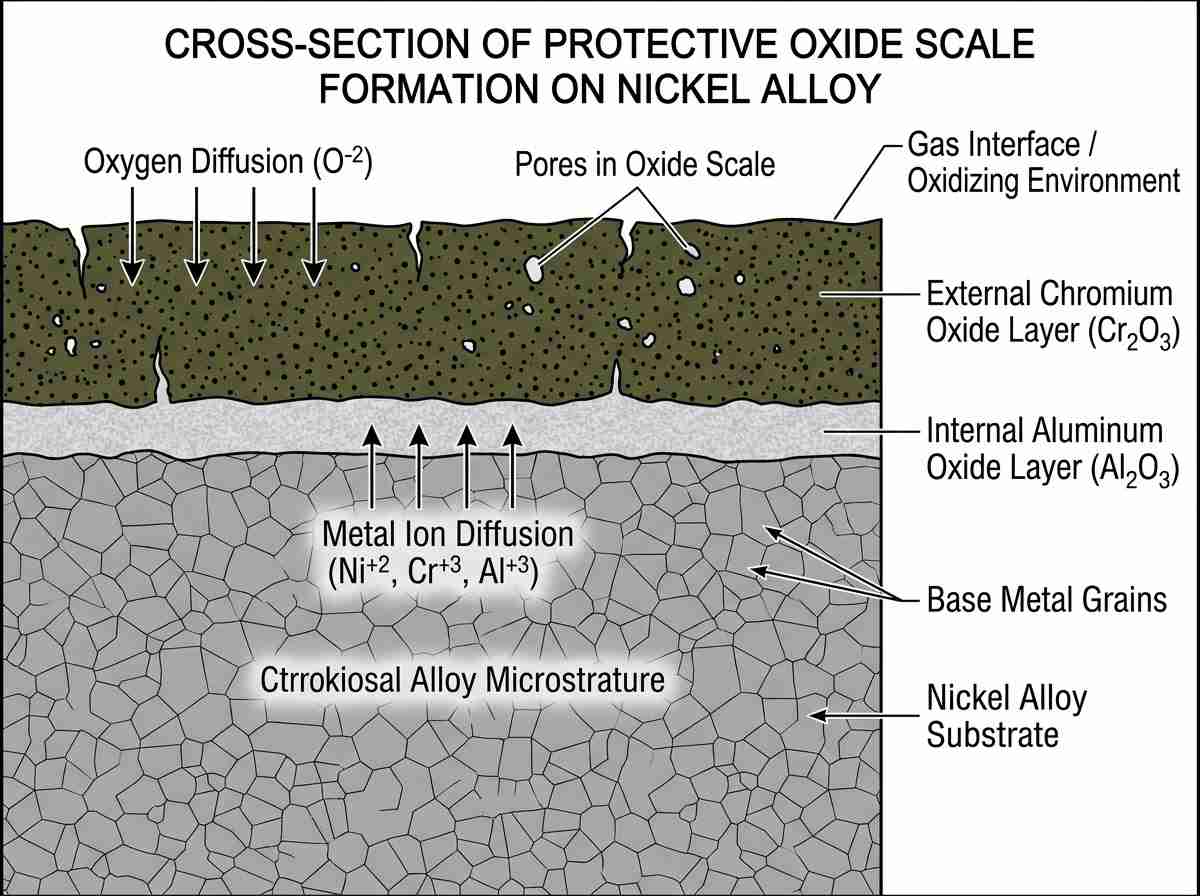
Another crucial factor limiting the nickel alloy working temperature range is environmental attack. While the matrix provides strength, the alloy relies on a continuous, adherent surface oxide scale—typically chromia (Cr2O3) or alumina (Al2O3)—for protection. Above 1000°C, chromia scales can volatilize into CrO3, rapidly accelerating material loss. For operations exceeding this threshold, alumina-forming alloys are mandatory.
In conclusion, relying on generic material data sheets is insufficient for critical engineering designs. Identifying the exact thermal limits requires a rigorous analysis of the specific operating environment, mechanical loads, and the intended lifespan of the component. If you are navigating complex material selection challenges and need to accurately define the ideal alloy for your application, reach out to the engineering team at 28Nickel for specialized technical support and metallurgical analysis.
Related Q&A
Q: Does the nickel alloy working temperature range shift under cryogenic conditions?
A: Yes. Unlike carbon steels that undergo a ductile-to-brittle transition, austenitic nickel alloys retain excellent toughness, ductility, and high yield strength at cryogenic temperatures (down to -196°C or even liquid helium temperatures). Their face-centered cubic (FCC) crystal structure prevents low-temperature embrittlement, expanding their total operational spectrum significantly.
Q: How does severe sulfur exposure alter the effective nickel alloy working temperature range?
A: Environments containing hydrogen sulfide (H2S) or sulfur dioxide (SO2) drastically reduce the safe maximum operating temperature. Nickel readily reacts with sulfur at elevated temperatures to form low-melting-point nickel sulfide eutectics (melting around 645°C). This causes rapid intergranular attack and catastrophic failure, necessitating alloys with higher chromium and lower iron/nickel ratios for high-temperature sulfidation resistance.
Q: Why does Inconel 718 have a lower maximum operating temperature than standard solid solution alloys?
A: Inconel 718 derives its immense strength from the metastable γ′′ (gamma double prime) phase. When the temperature exceeds 650°C, this phase thermally degrades, coarsening and transforming into the acicular (needle-like) δ phase. This transformation significantly depletes the matrix of strengthening elements, causing a rapid and irreversible loss of mechanical strength, defining its strict upper thermal limit.