Equipment failure in aggressive chemical processing environments is rarely just an inconvenience; it is often a catastrophic safety hazard and a massive financial drain. Engineers constantly grapple with selecting the optimal material to withstand pitting, crevice corrosion, and chloride-induced stress corrosion cracking (SCC). Conducting a precise nickel alloy corrosion resistance comparison is critical before finalizing any pressure vessel or piping specification. Different chemical media interact uniquely with specific alloying elements. Therefore, relying on generic material grades without a targeted nickel alloy corrosion resistance comparison often leads to premature degradation. In highly acidic environments, the synergistic effects of chromium, molybdenum, and nitrogen dictate the passivation behavior of the metal surface.
Key Metrics in Nickel Corrosion Environments
When evaluating a nickel alloy corrosion resistance comparison for reducing acids, we must look beyond basic tensile strength and focus on the Pitting Resistance Equivalent Number (PREN). A fundamental metric in any nickel alloy corrosion resistance comparison is this PREN value, calculated precisely as %Cr + 3.3(%Mo) + 16(%N). While PREN provides a theoretical baseline, a true nickel alloy corrosion resistance comparison requires examining the behavior of these materials at elevated temperatures and varying concentrations of corrosive media.
For instance, Alloy 600 (UNS N06600) offers excellent resistance to high-purity water and alkaline conditions but struggles in severe acidic environments compared to highly alloyed grades. By examining our internal nickel alloy corrosion resistance comparison data, the superiority of molybdenum-rich alloys like Hastelloy C-276 (UNS N10276) in localized corrosion environments becomes undeniable. C-276 exhibits exceptional resistance to wet chlorine gas, hypochlorite, and chlorine dioxide solutions. Conversely, Hastelloy B-3 is engineered specifically for hydrochloric acid at all concentrations and temperatures, but its performance drops significantly in oxidizing media. Thus, reviewing the nickel alloy corrosion resistance comparison table below is essential for matching the microstructure to the exact chemical service profile.
| Alloy Grade | UNS Number | Nominal Cr (%) | Nominal Mo (%) | PREN (Approx) | Best Service Environment |
| Alloy 400 | N04400 | 0 | 0 | 0 | Hydrofluoric acid, marine |
| Alloy 600 | N06600 | 15.5 | 0 | 15.5 | High purity water, dry Cl |
| Alloy 625 | N06625 | 21.5 | 9.0 | ~51 | Seawater, oxidizing acids |
| Alloy 825 | N08825 | 21.5 | 3.0 | ~31 | Sulfuric acid, phosphoric acid |
| Hastelloy C-276 | N10276 | 15.5 | 16.0 | ~68 | Wet chlorine, strong oxidizers |
Evaluating High-Temperature Acid Performance
To effectively utilize a nickel alloy corrosion resistance comparison, engineers must factor in the iso-corrosion curves for their specific operating temperatures. A typical benchmark is the 0.1 mm/year (4 mpy) corrosion rate contour line. Beyond purely theoretical data, an applied nickel alloy corrosion resistance comparison requires understanding the impact of trace impurities. Ferric or cupric ions in hydrochloric acid can unexpectedly shift the environment from reducing to oxidizing, rapidly accelerating the corrosion rate of Alloy B-3 while Alloy C-276 remains incredibly stable.
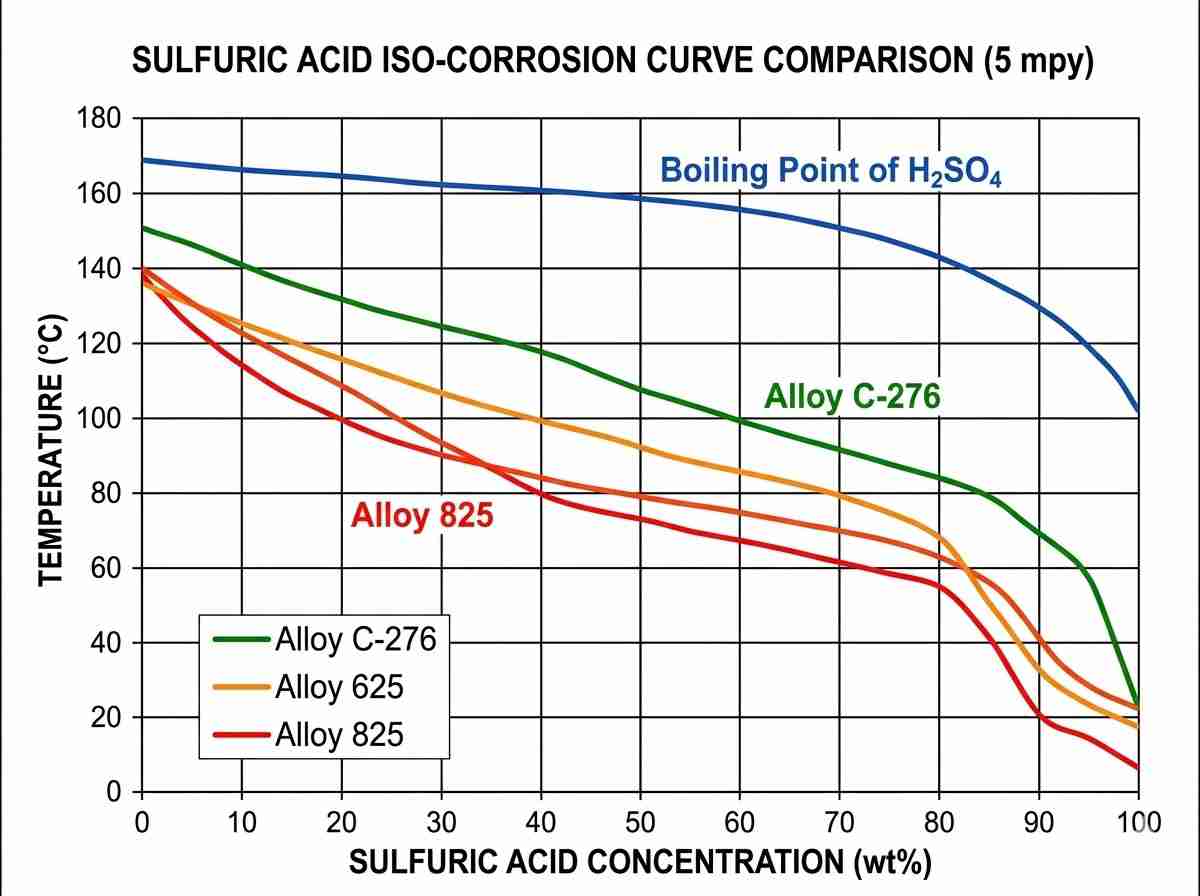
Furthermore, let’s consider harsh sulfuric acid applications. Alloy 825 is traditionally deployed here due to its specific copper addition, which significantly enhances resistance in reducing environments. However, as the concentration and temperature cross the 80°C threshold at 40% concentration, an in-depth nickel alloy corrosion resistance comparison will steer engineers toward Alloy G-30 or Alloy 625. These metallurgical transitions highlight exactly why a simple datasheet is never enough. The passive oxide layer’s stability is highly dependent on both temperature fluctuations and fluid velocity, parameters that must be modeled accurately in any rigorous analysis.
Alloy Stress Corrosion Cracking Resistance
Another vital dimension of evaluating these materials involves susceptibility to chloride-induced stress corrosion cracking (SCC). Standard austenitic stainless steels like 304 and 316 are notorious for SCC failure in hot chloride environments. By significantly increasing the base nickel content, these advanced alloys transition into a domain of near-immunity to chloride SCC. Alloy 625 and Alloy C-276, containing roughly 58% and 57% nickel respectively, provide this critical safeguard in offshore topside processing and downhole sour gas environments involving high partial pressures of H2S and CO2.
Moreover, thermal stability during structural welding is a paramount concern for fabricators. Sensitization, which is the precipitation of chromium carbides at the grain boundaries, can lead to severe intergranular corrosion in the heat-affected zone (HAZ). Our technical materials selection protocols consistently emphasize selecting low-carbon or titanium/niobium stabilized grades to mitigate this inherent risk. For example, the restricted carbon and silicon content in modern C-276 prevents grain boundary precipitates during welding, making it highly suitable for chemical process applications in the as-welded condition without requiring post-weld heat treatment.
Conclusion
Ultimately, specifying the correct pressure vessel or piping material demands far more than a cursory glance at chemical composition charts. It requires a rigorous, data-backed nickel alloy corrosion resistance comparison tailored to the precise thermodynamics and kinetics of your unique fluid system. Over-alloying drastically increases project capital expenditures without operational justification, while under-alloying invites catastrophic failure and facility downtime. For custom metallurgical analysis engineered strictly for your facility’s exact operational parameters, reach out to the materials engineering team at 28Nickel. We provide deep technical assessments, localized corrosion modeling, and metallurgical guidance to ensure your next critical deployment is engineered for maximum longevity and absolute safety.
Related Q&A:
Q1: Why is Alloy C-276 preferred over Alloy 625 in wet chlorine environments?
A: While both are highly alloyed, C-276 possesses a higher molybdenum content (16% vs 9%) and a deliberate addition of tungsten. This specific microstructure provides vastly superior resistance to localized pitting and crevice corrosion in highly oxidizing, chloride-rich media like wet chlorine gas.
Q2: Can we use Alloy B-3 in environments containing both hydrochloric and nitric acids?
A: No. Alloy B-3 is uniquely formulated for pure reducing environments, such as un-aerated hydrochloric acid. The presence of an oxidizing agent, such as nitric acid or even trace ferric ions, destroys its protective passive layer and leads to rapid, catastrophic uniform corrosion.
Q3: How does the PREN value accurately predict performance in real-world applications?
A: PREN (Pitting Resistance Equivalent Number) is purely an empirical calculation (%Cr + 3.3%Mo + 16%N) used to rank localized corrosion resistance. While excellent for initial screening, it does not account for operating temperature, fluid velocity, or synergistic chemical reactions, requiring iso-corrosion curves for final validation.