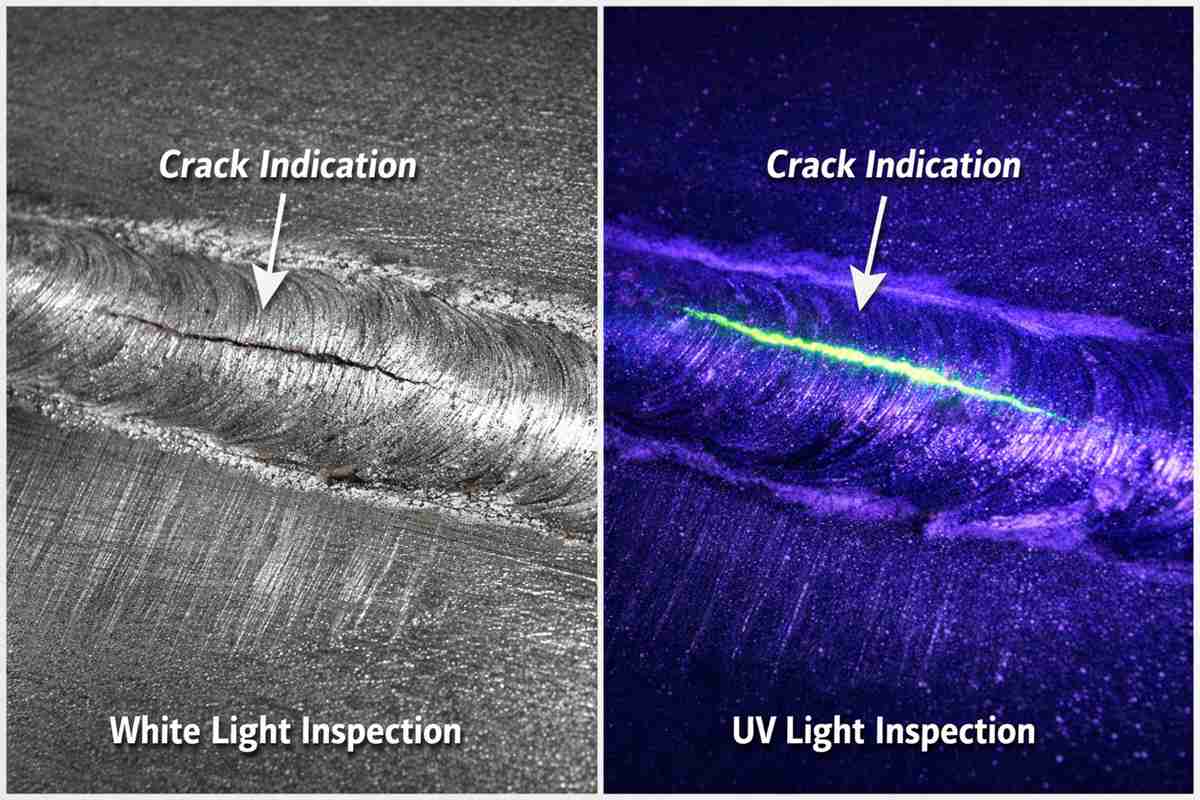
Evaluating the microstructural integrity of metallurgical components in aggressive chloride environments often reveals a glaring vulnerability: localized corrosion. For chemical processing, aerospace, and offshore extraction applications, specifying the correct barrier is an absolute necessity. When analyzing the broad spectrum of nickel alloy materials for sale, engineers frequently encounter baseline grades that lack the necessary molybdenum and tungsten additions to maintain long-term passivity. At 28Nickel, our failure analysis protocols consistently demonstrate that relying on standard austenitic stainless steels in sour gas or high-chloride media dramatically accelerates stress corrosion cracking (SCC) and pitting. Selecting the appropriate alloy demands a rigorous understanding of solid solution strengthening, phase stability, and environmental thermodynamics.

The primary metric dictating performance in these highly corrosive ecosystems is the Pitting Resistance Equivalent Number (PREN). This calculation is fundamental when evaluating any nickel alloy materials for sale intended for harsh service. By factoring in the mass fractions of chromium, molybdenum, and nitrogen, we can accurately predict a matrix’s resistance to localized anodic dissolution. For instance, while Alloy 825 provides adequate defense against mild sulfuric acid, it will rapidly falter in extreme pitting environments due to a significantly lower PREN compared to heavily alloyed counterparts.
When you critically analyze premium nickel alloy materials for sale designed for severe environments, the metallurgical focus shifts to superalloys where the synergistic effect of nickel, chromium, and molybdenum creates an almost impenetrable, self-repairing oxide layer. Furthermore, the inherently high nickel content in these specific grades effectively mitigates chloride-ion stress corrosion cracking—a catastrophic, sudden failure mode that plagues lesser alloys and causes unpredictable facility downtime. To understand the variance, we must look at the specific elemental breakdown.
| Alloy Grade | Nickel (Ni) % | Chromium (Cr) % | Molybdenum (Mo) % | Iron (Fe) % | Typical PREN | Primary Application Environment |
| Alloy 400 | 63.0 Min | – | – | 2.5 Max | N/A | Hydrofluoric acid, marine environments |
| Alloy 600 | 72.0 Min | 14.0 – 17.0 | – | 6.0 – 10.0 | ~15 | High-temperature oxidation, dry chlorine |
| Alloy 825 | 38.0 – 46.0 | 19.5 – 23.5 | 2.5 – 3.5 | 22.0 Min | ~31 | Phosphoric acid, moderate sour gas |
| Alloy 625 | 58.0 Min | 20.0 – 23.0 | 8.0 – 10.0 | 5.0 Max | ~51 | Severe localized corrosion, high-stress |
| Alloy C-276 | Balance | 14.5 – 16.5 | 15.0 – 17.0 | 4.0 – 7.0 | ~68 | Wet chlorine gas, hypochlorites |
Beyond localized aqueous corrosion, high-temperature structural stability is equally paramount in material selection. Creep deformation occurs when metallurgical bonds are subjected to continuous mechanical stress at elevated temperatures, typically above 0.4 times the absolute melting temperature of the metal. If your project requires high-grade nickel alloy materials for sale, analyzing the precipitation hardening phases, such as gamma prime and gamma double prime intermetallics, is strictly non-negotiable.
These microscopic precipitates effectively pin dislocation movements within the crystal lattice, drastically reducing creep rates and extending the component’s operational lifespan. Furthermore, the strategic inclusion of trace elements like niobium in specific nickel alloy materials for sale enhances structural weldability while simultaneously preventing strain-age cracking during post-weld heat treatment (PWHT). It is this delicate balance of elemental additions that separates standard materials from true high-performance superalloys.
Ultimately, standardizing your material specifications requires moving far beyond generic datasheets and basic tensile strength figures. The complex metallurgical variances between different nickel alloy materials for sale directly and immediately impact your equipment’s lifecycle, maintenance intervals, and overall operational safety. At 28Nickel, our engineering team focuses heavily on matching the precise phase thermodynamics and mechanical properties of our alloys to your highly specific operational parameters. By thoroughly understanding the limits of your current infrastructure, we can engineer a metallurgical solution that eliminates premature yield failures. We strongly encourage engineers and metallurgists dealing with aggressive media to share their operational data, thermal profiles, and system schematics with our technical team to determine the exact microstructural requirements for their upcoming projects.
Related Q&A:
Q1: Why do specific nickel alloy materials for sale outperform standard stainless steel in sour gas applications?
A1: Sour gas environments contain high concentrations of hydrogen sulfide (H2S). The significantly elevated nickel and molybdenum content in specialized superalloys stabilizes the austenite phase and prevents both hydrogen embrittlement and sulfide stress cracking (SSC), which rapidly degrade standard 300-series stainless steels.
Q2: How does elevated operating temperature affect the PREN requirements for nickel alloy materials for sale?
A2: Higher temperatures exponentially increase the reactivity and penetration capability of chloride ions. Therefore, nickel alloy materials for sale operating in aqueous environments above 150°C require a significantly higher PREN—typically exceeding 45—to maintain the Critical Pitting Temperature (CPT) and preserve the protective passive oxide layer.
Q3: Can the underlying crystal lattice structure of nickel alloy materials for sale degrade over time?
A3: Yes, prolonged thermal exposure to temperatures outside the alloy’s specifically designed thermal envelope can cause detrimental phase precipitation, such as sigma or mu phase formation, which drastically reduces impact toughness. Selecting the properly stabilized nickel alloy materials for sale ensures long-term microstructural stability throughout the component’s intended lifecycle.