Hastelloy C-22 corrosion resistance in sulfuric acid is often discussed as if it were a single, fixed property. In real plant service, it is nothing of the sort. The performance of UNS N06022 shifts with acid concentration, temperature, oxidizing contamination, flow regime, and whether the data comes from clean laboratory acid or a dirty process loop carrying chlorides, ferric ions, or dissolved oxygen. That distinction matters, because many costly material failures happen when buyers treat “good sulfuric acid resistance” as a universal claim instead of a concentration-temperature envelope. HASTELLOY C-22 is a Ni-Cr-Mo-W alloy with a nominal chemistry of Ni balance, 22% Cr, 13% Mo, 3% W, and 3% Fe, and its high chromium content is one reason it performs well not only in reducing media but also in oxidizing or mixed-acid environments.
From a corrosion engineer’s perspective, Hastelloy C-22 corrosion resistance in sulfuric acid should be judged less by brochures and more by three questions: what is the acid concentration, what is the true metal temperature, and what impurities are present? Haynes’ corrosion data and iso-corrosion diagrams are based on reagent-grade sulfuric acid under laboratory conditions, and Haynes explicitly recommends field testing before industrial use. That caveat is not legal fine print; it is the core of sound alloy selection.
Why Hastelloy C-22 Corrosion Resistance in Sulfuric Acid Is Not a Single Number
The metallurgy explains the behavior. In pure sulfuric acid, molybdenum is highly beneficial, and nickel-molybdenum alloys generally show the strongest resistance. But industrial sulfuric acid is rarely an ideal reducing medium. Oxidizing species can destabilize alloys that look excellent in “clean acid” charts. Nickel-chromium-molybdenum alloys such as C-22 retain high sulfuric acid resistance while gaining extra protection from chromium when oxidizing species appear in service. Haynes’ broader corrosion guide states this directly: in industrial sulfuric acid containing oxidizing species, higher chromium within the Ni-Cr-Mo family increases protection. That is why C-22 is often selected not because it is the absolute best in pure sulfuric acid at every point, but because it is one of the most forgiving alloys in mixed, contaminated, or upset conditions.
This is also where engineers sometimes confuse C-22 with C-276. HASTELLOY C-22 has higher chromium than C-276 and is specifically noted by Haynes as having much higher resistance to oxidizing media and exceptional resistance to chloride-induced pitting. In sulfuric acid systems where chloride ingress, oxidizing carryover, or cleaning-cycle crossover can occur, that extra margin is often more valuable than chasing the best pure-acid number on paper. That is an engineering inference drawn from the published alloy chemistry and corrosion guidance, not a blanket rule for every plant.
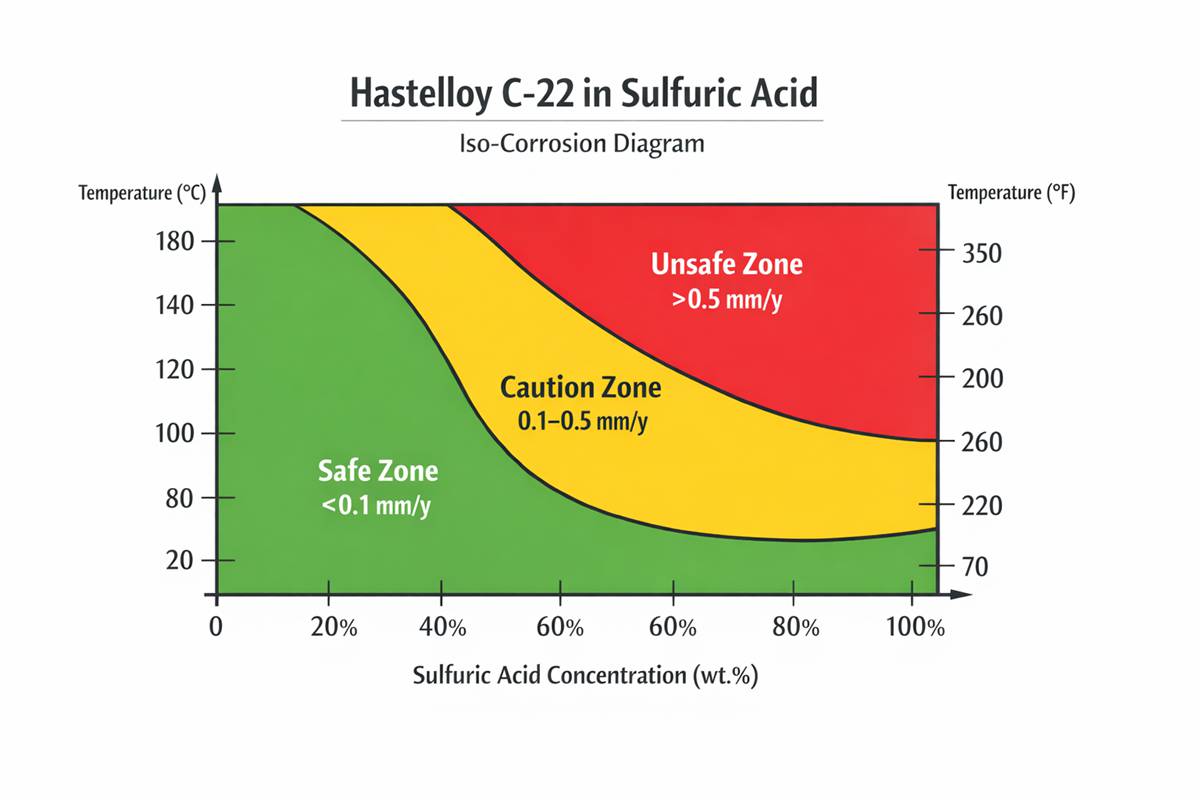
Below are representative laboratory corrosion rates for Hastelloy C-22 corrosion resistance in sulfuric acid, extracted from Haynes sulfuric-acid data for reagent-grade solutions. Lower numbers are better; as a practical screen, the Haynes iso-corrosion plots use 0.1 mm/y as a “very safe” threshold and 0.5 mm/y as a higher but sometimes still manageable boundary, depending on design life and corrosion allowance.
| Sulfuric Acid Concentration | 150°F / 66°C | 175°F / 79°C | 200°F / 93°C | Hervir |
|---|---|---|---|---|
| 10 wt.% | — | 0.02 mm/y | 0.04 mm/y | 0.29 mm/y |
| 20 wt.% | 0.01 mm/y | 0.03 mm/y | 0.28 mm/y | 0.83 mm/y |
| 40 wt.% | 0.01 mm/y | 0.31 mm/y | 0.87 mm/y | 3.99 mm/y |
| 60 wt.% | — | 0.67 mm/y | 0.95 mm/y | — |
| 80 wt.% | — | 1.44 mm/y | 2.16 mm/y | — |
| 96 wt.% | 0.10 mm/y | — | 1.10 mm/y | — |
A few patterns jump out immediately. First, Hastelloy C-22 corrosion resistance in sulfuric acid is strong at low to moderate concentrations when metal temperature is controlled. At 10 to 20 wt.% acid, the alloy remains in a very low corrosion regime up to roughly 79°C, but the rate climbs as temperature rises toward 93°C and above. Second, the mid-to-high concentration range is where the design window narrows fast. At 40 wt.% sulfuric acid, the rate is still only 0.01 mm/y at 66°C, yet it rises to 0.87 mm/y by 93°C and nearly 4 mm/y at boiling. Third, very concentrated acid is not automatically “safer.” At 96 wt.% sulfuric acid, C-22 is about 0.10 mm/y at 66°C, but already about 1.10 mm/y at 93°C.
How to Use the Data in Real Equipment
For reactors, acid coolers, absorber piping, transfer spools, and tower internals, Hastelloy C-22 corrosion resistance in sulfuric acid should be validated against the actual metal temperature, not only bulk liquid temperature. I have seen acceptable lab numbers misapplied to steam-jacketed nozzles, dead legs, weld HAZ zones under deposits, and pump discharge sections where flash heating changed the corrosion regime. In sulfuric acid service, ten or fifteen degrees can move an alloy from comfortably below 0.1 mm/y into a rate that begins to erode maintenance intervals. The alloy data tells you the direction of risk; your process details determine how fast you arrive there.
Another point that deserves emphasis: laboratory sulfuric acid data does not fully capture contaminated plant acid. The Haynes corrosion guide notes that highly concentrated industrial sulfuric acid, especially 92 to 99 wt.% acid produced from smelter off-gases, behaves as a “super-oxidizing” environment. In those solutions, nickel-chromium-molybdenum alloys may still be usable up to about 95°C, but above that temperature other material systems, including high-silicon nickel-based or stainless materials, may be required. That matters for acid concentration plants, gas-cleaning trains, and heat-recovery circuits where buyers assume a Ni-Cr-Mo alloy is automatically the end of the discussion. It is not.
So, is Hastelloy C-22 a good choice for sulfuric acid? Yes—often an excellent one—but only when the selection logic is disciplined. For clean, purely reducing sulfuric acid, some higher-molybdenum alloys can outperform it over wider temperature windows. For sulfuric acid streams that may carry oxidizers, chlorides, or mixed chemistry, C-22 is frequently the more balanced choice because chromium improves robustness against those real-world complications. That conclusion is consistent with the published corrosion guidance, though final selection still requires service-specific review.

Conclusión
If your team is evaluating Hastelloy C-22 corrosion resistance in sulfuric acid, do not stop at a generic alloy datasheet. Start with concentration, true wall temperature, oxidizing impurities, chloride contamination, velocity, and whether the component sees stagnant pockets or weld-adjacent heat tint. That is the difference between buying an expensive alloy and buying the right alloy. If you can share your acid concentration range, operating and upset temperatures, impurity profile, and expected equipment life, a proper corrosion review can usually narrow the material choice very quickly.
Preguntas y respuestas relacionadas
1) Is Hastelloy C-22 better than 316L in sulfuric acid?
In most meaningful sulfuric acid duties, yes. Haynes’ comparative corrosion information shows nickel-based corrosion-resistant alloys operating over much broader sulfuric-acid temperature windows than common austenitic stainless steels. For anything beyond mild, low-temperature exposure, 316L is usually not the serious benchmark.
2) Can Hastelloy C-22 handle concentrated sulfuric acid above 90%?
It can in some ranges, but not without temperature discipline. Published C-22 data shows about 0.10 mm/y at 96 wt.% sulfuric acid and 66°C, but about 1.10 mm/y at 93°C. In highly concentrated industrial acid, the chemistry can become strongly oxidizing, so plant-specific validation is essential.
3) What information should a supplier review before recommending C-22 for sulfuric acid service?
At minimum: acid concentration range, operating and upset temperature, impurities such as chlorides or ferric ions, aeration level, flow velocity, erosion risk, weld condition, and target service life. Haynes also explicitly advises field testing because lab data is generated in reagent-grade acids under controlled conditions.