When engineers ask about Incoloy 825 corrosion resistance in sulfuric acid, the right answer is not a simple yes or no. Alloy 825 is one of the more dependable nickel-iron-chromium grades for sulfuric acid duty, but its behavior changes sharply with acid concentration, temperature, aeration, and contamination. In practice, that is exactly why some plants get long service life from 825 while others see rapid thinning, weld-area attack, or upset-driven corrosion after what looked like a “safe” material selection on paper.
From a metallurgy standpoint, 825 was built for mixed corrosive environments rather than for one narrow condition. Its nickel base helps suppress chloride stress-corrosion cracking; chromium supports passivation in oxidizing conditions; molybdenum improves resistance to localized attack; copper is especially useful in reducing acids such as sulfuric acid; and titanium stabilization helps the alloy resist sensitization-related intergranular attack in the welded condition. That combination is why Alloy 825 has remained relevant in acid handling, pickling systems, scrubbers, and chemical process equipment for decades.
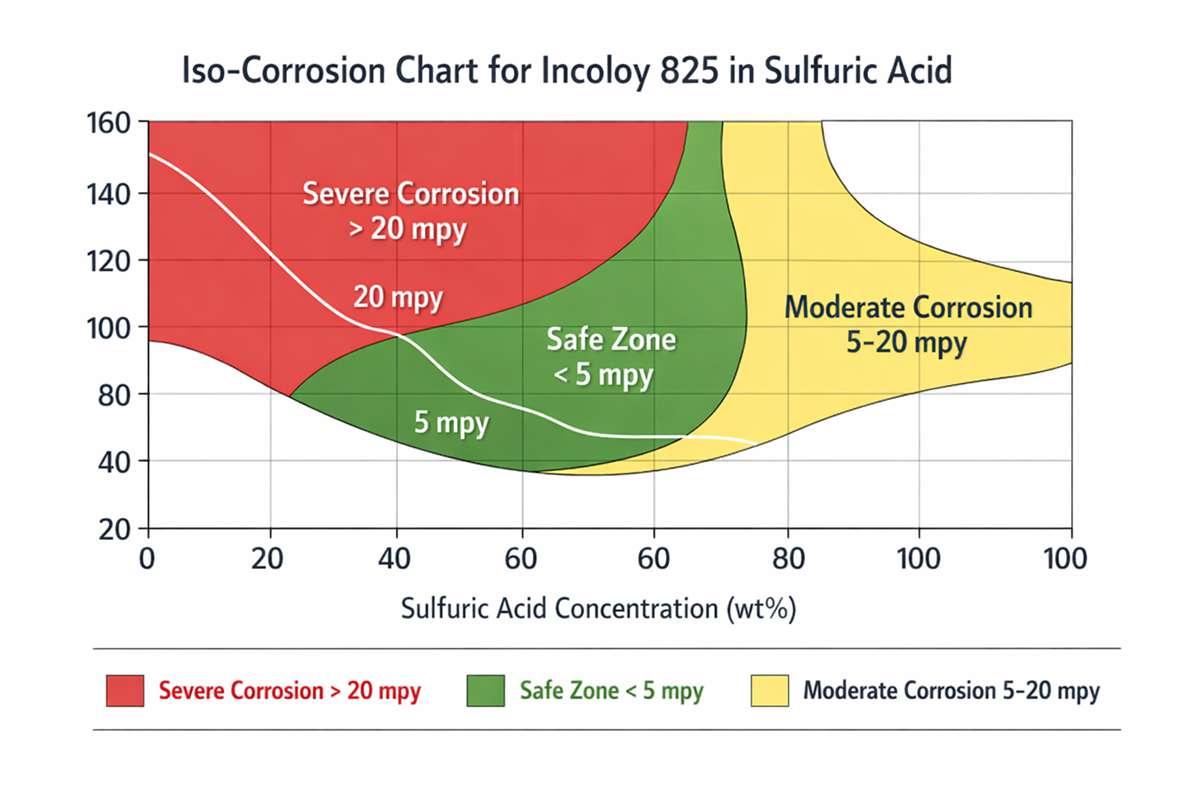
How Incoloy 825 performs in sulfuric acid
The key point is that sulfuric acid is not a uniform environment. Dilute sulfuric acid is strongly reducing. As concentration and temperature shift, the corrosion mechanism also shifts. Incoloy 825 generally performs best in what engineers often call the intermediate concentration window, where the alloy can maintain a more stable surface condition. Special Metals reports very good resistance, defined there as below 5 mpy (below 0.13 mm/a), in sulfuric acid between roughly 40 and 80 wt% at 50°C under representative service conditions. That is excellent screening-level performance, but it should never be treated as a blanket guarantee for every plant stream.
This is also why experienced corrosion engineers do not select 825 by concentration alone. A nominal “50% sulfuric acid” stream in a real unit may carry ferric ions, chlorides, dissolved copper salts, suspended solids, or oxygen fluctuations. Those details matter. The same Special Metals guidance notes that severe departures from expected behavior can occur in contaminated intermediate-strength acid. It also notes something many non-specialists miss: some oxidizing salts can actually help, while chlorides are notoriously harmful in sulfuric acid service. In continuously submerged chloride-contaminated duty, higher-molybdenum alloys such as C-276, 625, or 686 may be required.
A useful sanity check comes from boiling laboratory sulfuric acid data. In one published comparison, Alloy 825 showed corrosion rates of about 20 mpy at 10% sulfuric acid, 11 mpy at 40%, and 20 mpy at 50%, while 316 stainless steel performed dramatically worse, reaching 636 mpy at 10% and over 1000 mpy at both 40% and 50%. The lesson is not that 825 is universally “good” at all boiling concentrations. The real lesson is that 825 occupies a very different corrosion class from conventional stainless steels in sulfuric acid, especially once the medium becomes more aggressive.
Practical selection table for Incoloy 825 in sulfuric acid
| Sulfuric acid condition | Typical reading for Alloy 825 | Engineering interpretation | Material selection note |
|---|---|---|---|
| Dilute acid at elevated temperature | Performance can deteriorate quickly | Reducing conditions are severe; passivation is harder to maintain | Verify with corrosion test data before committing |
| Intermediate concentrations, moderate temperature | Often the strongest operating window | 825 is well known for stable behavior here | Commonly suitable for tanks, piping, pumps, and heat-exchanger parts |
| Boiling laboratory acid at 10% | ~20 mpy (0.5 mm/a) | Not catastrophic, but not “ignore it” corrosion | Allowance or upgrade may be needed |
| Boiling laboratory acid at 40% | ~11 mpy (0.28 mm/a) | Better than many stainless grades by a wide margin | Often acceptable with proper design review |
| Boiling laboratory acid at 50% | ~20 mpy (0.5 mm/a) | Still workable in some cases, but not a universal answer | Check upset conditions and impurities carefully |
| Chloride-contaminated sulfuric acid | Risk rises sharply | Localized attack and loss of expected life are common failure drivers | Consider higher-Mo alloys if chlorides are persistent |
| Welded equipment | Usually reliable when properly processed | Titanium stabilization helps reduce sensitization risk | Use qualified filler/heat input control and review post-fabrication condition |
Note: The table combines published laboratory sulfuric acid data for Alloy 825 with vendor iso-corrosion guidance and should be used for preliminary selection, not final design approval.
Limits of Incoloy 825 corrosion resistance in sulfuric acid service
The biggest selection mistake is assuming that 825 is a “sulfuric-acid-proof” alloy. It is not. It is a broad-duty corrosion alloy with a particularly useful window in sulfuric acid. When the process becomes hotter, more dilute than expected, chloride-bearing, or subject to concentration cycling, metal loss can move from manageable to unacceptable faster than many buyers anticipate. This is especially true in areas with stagnant flow, gasket-shadowed crevices, dead legs, and partially wetted vapor-liquid interfaces, where the local chemistry may be much harsher than the bulk analysis suggests.
There is also a commercial point here that engineers and procurement teams should align on early: the cheapest winning option is not always 825, and the safest option is not always a higher alloy. If the duty is relatively clean sulfuric acid in the favorable concentration-temperature range, 825 can be a very rational balance of corrosion resistance, fabricability, and cost. If chlorides are sustained, or if upset conditions push the unit outside that window, the more expensive upgrade can still be the lower life-cycle-cost decision. Special Metals even notes that Alloy 20 behaves similarly in sulfuric acid service in some comparable applications, which means a serious material review should usually compare 825, Alloy 20, and higher-Mo nickel alloys rather than defaulting to one family.
Welding, fabrication, and field reality
For fabricated equipment, the corrosion story is never only about the base metal. Alloy 825 is readily formed and welded, and its titanium stabilization is one reason it resists intergranular attack after thermal exposure that would sensitize unstabilized stainless steels. That matters in sulfuric acid systems because many real failures start not in the plate center but at weld toes, heat-affected zones, attachment points, or residual-stress concentrators. Good filler selection, controlled heat input, clean pickling/passivation practice where applicable, and avoidance of sulfur-bearing shop contamination all improve the odds that the installed equipment behaves like the datasheet, not like the failure report.

Conclusion
So, is Incoloy 825 corrosion resistance in sulfuric acid good? Yes, often very good, but only inside a definable operating window. The alloy is strongest where sulfuric acid concentration and temperature allow a stable surface condition, and it loses margin when chloride contamination, boiling dilute acid, crevice geometry, or process upsets dominate the corrosion mechanism. That is why good material selection for sulfuric acid service is never just a grade name on a PO. It is a media review.
If you are evaluating plate, pipe, tube, bar, or forged components for sulfuric acid duty, 28Nickel can help you compare 825 with Alloy 20, 625, C-276, and other options against your actual concentration-temperature-contamination envelope, not just nominal process data. In sulfuric acid service, that difference is often what separates a long campaign from an early shutdown.
Related Q&A
1. Is Incoloy 825 better than 316 stainless steel in sulfuric acid?
In most meaningful sulfuric acid duties, yes. Published boiling laboratory data show Alloy 825 far outperforming 316 stainless steel, especially at 40% and 50% sulfuric acid, where 316 degrades very rapidly.
2. What is the biggest risk factor for Alloy 825 in sulfuric acid service?
Unexpected chlorides are high on the list. Chloride contamination can sharply reduce corrosion resistance and may push the duty into a range where higher-molybdenum nickel alloys are the safer choice.
3. Can welded Alloy 825 be used in sulfuric acid equipment?
Usually yes, provided fabrication is done correctly. The alloy is stabilized with titanium to resist sensitization-related intergranular attack, but weld procedure control and service chemistry still matter.



