When engineers ask about Inconel 617 corrosion resistance in sulfuric acid, the honest answer is not a marketing slogan. It is a materials-selection discussion. Alloy 617 is a nickel-chromium-cobalt-molybdenum alloy best known for high-temperature strength, oxidation resistance, and metallurgical stability in severe thermal service. Those strengths are real. But sulfuric acid is not a single environment. Its aggressiveness changes sharply with concentration, temperature, aeration, contaminants, flow regime, and whether the alloy surface remains passive or is forced into active dissolution. That is where experienced selection work matters.
A common mistake is to assume that because Alloy 617 performs well in hot oxidizing gas, it will automatically be reliable in wet sulfuric acid. In practice, the corrosion mechanism is different. Dry high-temperature oxidation and aqueous acid corrosion are driven by very different surface chemistries. In sulfuric acid, especially under reducing or hot dilute conditions, the protective film can become unstable, and the alloy may shift from mild attack to unacceptable general corrosion far faster than many procurement specifications suggest.
Why Sulfuric Acid Is a Difficult Medium for Nickel Alloys
Sulfuric acid is deceptively complex. At first glance, buyers often reduce the question to concentration alone. That is too simplistic. In real plants, sulfuric acid service is shaped by at least six coupled variables: acid concentration, operating temperature, oxidizing species, halide contamination, fluid velocity, and fabrication condition.
Nickel provides a useful base in many corrosive environments, yet sulfuric acid is often more demanding than hydrochloric acid is in terms of passive film stability for certain nickel-chromium alloys. Chromium can help build passivity under the right electrochemical conditions; molybdenum can improve resistance to localized attack and some reducing media. But neither effect should be treated as absolute. Once the solution becomes hotter, more reducing, more contaminated, or more turbulent, the corrosion response can change abruptly.
For Alloy 617 specifically, this leads to an important engineering conclusion: Inconel 617 corrosion resistance in sulfuric acid is conditional, not universal. It may be acceptable in selected, tightly controlled cases. It is not the first-choice wet-process sulfuric acid alloy across the whole operating envelope.
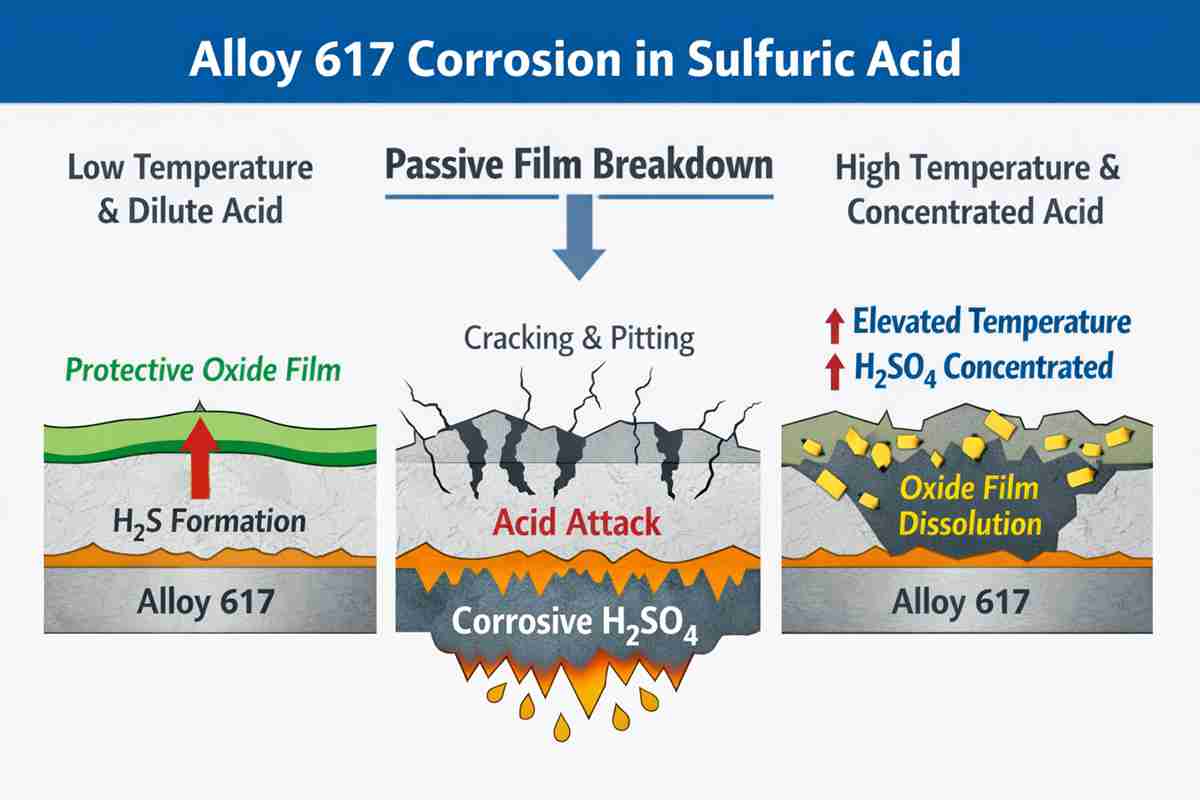
How Inconel 617 Behaves in Sulfuric Acid Service
Alloy 617 contains high nickel with significant chromium, cobalt, and molybdenum additions. That chemistry gives it impressive thermal capability, but sulfuric acid performance must still be judged case by case. In moderate environments, particularly where temperature is low and the chemistry is stable, the alloy can show usable resistance. Once temperature rises, however, or when acid remains dilute enough to stay strongly active, general corrosion can accelerate.
This is why seasoned corrosion engineers rarely approve Alloy 617 for sulfuric acid service based on composition alone. They ask harder questions. Is the acid clean or contaminated with chlorides, ferric ions, fluorides, or process carryover? Is the service continuous, or does shutdown create condensation and acid concentration cycling? Are there welds with heat tint left in place? Is the equipment seeing stagnant crevices or flashing flow? These details often determine whether the alloy survives or becomes an expensive lesson.
Another nuance is often overlooked. Alloy 617 may perform very well in hot gas zones of sulfur-bearing equipment, but when the process cools below the acid dew point and condensed sulfuric acid forms, the selection logic changes. A material that is excellent above the dew point can become vulnerable below it. That transition zone deserves separate review.
Practical Screening Guide for Inconel 617 in Sulfuric Acid
The table below is not a substitute for plant-specific testing, but it is a realistic preselection framework engineers can use before moving to coupon trials or a formal corrosion review.
| Sulfuric acid condition | Expected behavior of Alloy 617 | Likely corrosion risk | Engineering comment |
|---|---|---|---|
| Very dilute acid at ambient temperature, clean system | Often conditionally acceptable | Mild to moderate general corrosion | Use only after verifying actual concentration control and surface condition |
| Dilute acid at elevated temperature | Frequently unfavorable | Rapid increase in uniform attack | Hot dilute sulfuric acid is one of the most dangerous assumptions for Alloy 617 |
| Intermediate concentration, moderate temperature | Unstable; highly chemistry-dependent | General corrosion, crevice attack | Small chemistry shifts can change performance sharply; testing is strongly advised |
| Concentrated acid at low temperature with tight water control | Sometimes more stable than hot dilute acid, but not automatically safe | Local attack if water ingress or contamination occurs | Do not generalize from dry or near-dry conditions to wet operating upset conditions |
| Acid with chlorides, fluorides, or oxidizing/reducing contaminants | Unpredictable without data | Localized attack, weld-zone sensitivity | Impurity profile can dominate alloy behavior more than nominal acid concentration |
| Welded components with heat tint or rough surface finish | Lower reliability than base metal | Preferential attack at weld and HAZ | Post-weld cleaning and representative welded coupon testing are essential |
A useful way to read this table is to focus less on the word “acceptable” and more on the phrase “under control.” The narrower the operating window, the less margin the alloy has. For sulfuric acid duty, margin matters.
What Engineers Should Verify Before Specifying Alloy 617
If you are evaluating Inconel 617 corrosion resistance in sulfuric acid for heat exchangers, duct transitions, acid dew-point zones, reactor internals, or fabricated vessels, ask for more than a generic corrosion allowance. Request process-specific inputs.
First, define the real chemistry, not the nominal chemistry. Plants rarely operate with perfectly clean sulfuric acid. Trace chlorides, iron salts, catalyst fines, sulfur oxides, or cleaning residues can push a borderline alloy into failure. Second, define the temperature profile, including startup, shutdown, and upset conditions. Many failures occur during transient conditions, not steady state. Third, include fabricated details. Base metal behavior is only part of the story; welded coupons, crevice assemblies, and surface-finished specimens give far more useful evidence.
From a testing standpoint, immersion data alone may be insufficient. Depending on the service, you may need welded coupon testing, flowing-loop exposure, or dew-point corrosion simulation. For critical equipment, the cheapest test is still far less expensive than one premature shutdown.
So, is Inconel 617 corrosion resistance in sulfuric acid good? In a narrow sense, sometimes yes. In a broad specification sense, that is the wrong question. The better question is whether Alloy 617 is the right alloy for your exact sulfuric acid regime. For many wet sulfuric acid applications, engineers may ultimately compare it against more acid-focused nickel alloys or other corrosion systems. But if your service combines high temperature, complex thermal cycling, and sulfur-bearing process exposure, Alloy 617 still deserves a serious technical review rather than a quick yes-or-no answer.
At 28Nickel, this is exactly where technical support adds value. A sound recommendation starts with service chemistry, fabrication details, and failure mode expectations—not just an alloy name on a purchase order.

Final Takeaway
Inconel 617 corrosion resistance in sulfuric acid should be treated as a controlled-use topic, not a blanket advantage claim. Alloy 617 brings strong nickel-alloy fundamentals and excellent high-temperature capability, but sulfuric acid service rewards precision. If the medium is hot, dilute, contaminated, stagnant, or condensation-prone, the risk rises quickly. Engineers and buyers who define the real service envelope early will make better alloy decisions and avoid costly field corrections.
If your team is screening materials for sulfuric acid duty, the fastest path is to review the concentration-temperature map, impurity profile, welded condition, and expected upset scenarios before final procurement.
Related Q&A
1. Is Inconel 617 better than 316L stainless steel in sulfuric acid?
Usually, yes in terms of overall nickel-alloy capability, especially where temperature or mixed corrosive conditions are severe. But that does not mean Alloy 617 is automatically suitable for hot or reducing sulfuric acid service.
2. Can welded Inconel 617 be used in sulfuric acid equipment?
It can be used, but welded condition must be evaluated carefully. Heat tint, surface roughness, weld-metal chemistry, and heat-affected-zone behavior can all reduce corrosion reliability if post-weld cleaning is poor.
3. What technical data should a buyer provide before ordering Alloy 617 for sulfuric acid service?
At minimum: acid concentration range, normal and upset temperature, velocity, impurities, presence of chlorides or fluorides, aeration level, shutdown/condensation risk, and whether the part is welded or formed. Without those details, alloy selection is guesswork.