Quando gli ingegneri chiedono informazioni su Hastelloy C-276 resistenza alla corrosione in acido solforico, they are usually not asking a theoretical question. They are trying to stop a very practical failure: leaking transfer lines, exchanger plate loss, unexpected weld attack, or a vessel that looked acceptable on a generic corrosion chart but did not survive real liquor chemistry. C-276 remains one of the most broadly specified Ni-Cr-Mo alloys because its chemistry balances reducing-acid capability with a meaningful tolerance for oxidizing contaminants. But in sulfuric acid service, the correct answer is never just “excellent.” It depends on concentration, temperature, aeration, and what else is dissolved in the acid.
At the metallurgy level, that balance is not accidental. Haynes lists C-276 at roughly balance nickel, 16 wt.% chromium, 16 wt.% molybdenum, and 4 wt.% tungsten, with extremely low carbon and silicon. In practice, that is why the alloy can handle both oxidizing and non-oxidizing acids better than most stainless grades, while also keeping welded zones far less vulnerable than older corrosion alloys. For sulfuric acid systems, this combination matters because the acid can swing from predominantly reducing behavior to more complex electrochemistry as concentration rises, especially around the 60 to 70 wt.% range.
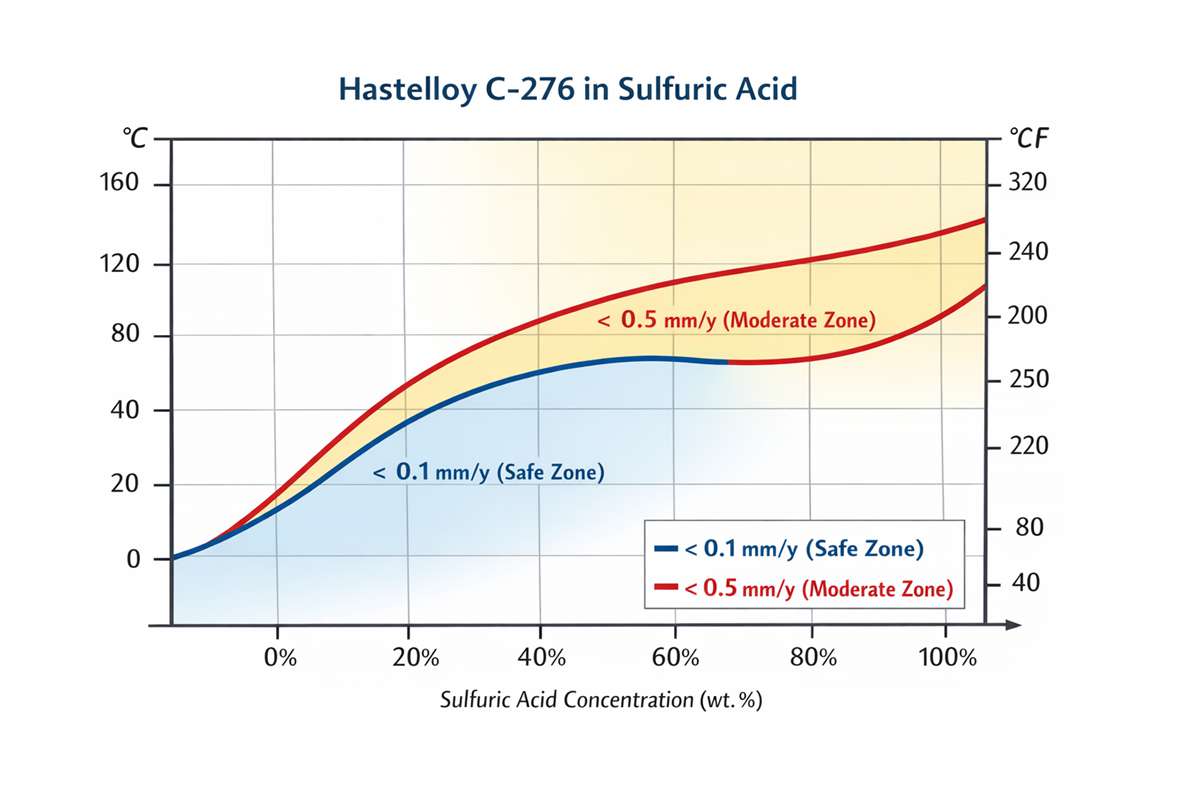
The most useful way to read Hastelloy C-276 corrosion resistance in sulfuric acid is through iso-corrosion logic rather than a single “yes/no” material rating. Haynes defines the blue line on its diagrams as 0.1 mm/y and the red line as 0.5 mm/y. Below the 0.1 mm/y line, corrosion is generally considered very safe for long-life equipment; between 0.1 and 0.5 mm/y, the alloy can still be viable depending on corrosion allowance, inspection interval, and component criticality. On the comparative sulfuric-acid plot, C-276 sits clearly above 316L, 254SMO, and alloy 625 over much of the concentration range, which explains why it is so often selected for aggressive CPI sulfuric service when stainless steels become marginal.
A few actual data points tell the story better than marketing language:
| Sulfuric acid concentration (wt.%) | 66°C / 150°F (mm/y) | 79°C / 175°F (mm/y) | 93°C / 200°F (mm/y) | 107°C / 225°F (mm/y) | 121°C / 250°F (mm/y) | Boiling (mm/y) |
|---|---|---|---|---|---|---|
| 10 | — | 0.03 | 0.14 | — | — | 0.18 |
| 20 | — | 0.05 | 0.40 | — | — | 0.49 |
| 50 | 0.02 | 0.26 | 0.62 | 1.13 | 2.33 | 3.64 |
| 70 | 0.05 | 0.16 | 0.50 | 1.06 | 13.68 | — |
| 90 | 0.03 | 0.05 | 0.46 | 1.64 | 4.79 | — |
| 96 | — | 0.04 | 0.18 | 0.95 | — | — |
Source note: reagent-grade sulfuric acid, laboratory conditions; Haynes explicitly recommends field testing before industrial use.
What jumps out is that Hastelloy C-276 corrosion resistance in sulfuric acid is not controlled by concentration alone. At 70 wt.% acid, the alloy still shows only 0.16 mm/y at 79°C and 0.50 mm/y at 93°C, but it climbs sharply to 13.68 mm/y at 121°C. At 90 wt.% acid, the same alloy looks surprisingly comfortable at 79°C, then moves beyond a conservative long-life regime as temperature rises. In other words, temperature is usually the first variable to pin down, not the last. That is exactly why engineers get into trouble when procurement only asks for “sulfuric acid concentration” and ignores the real thermal envelope.
There is another practical twist. Pure sulfuric acid data are useful, but plant acid is often not pure. Haynes notes that Ni-Cr-Mo alloys such as C-276 retain high resistance in pure sulfuric acid, while chromium also gives some protection against oxidizing species present in industrial solutions. At the same time, those same industrial solutions can become much less forgiving than reagent-grade acid. In highly concentrated industrial sulfuric acid, especially the 92 to 99 wt.% “super-oxidizing” range from smelter off-gases, nickel-chromium-molybdenum alloys can still be used up to about 95°C, but above that temperature other materials that form alternate protective films may be required. That boundary is where a generic corrosion chart stops being enough.
For fabricators, the welding point is equally important. One reason buyers continue to specify C-276 for vessels, nozzles, reducers, and spool assemblies is that its very low carbon and silicon help preserve corrosion resistance after welding. Haynes’ weld-corrosion data in sulfuric acid at 66°C show all-weld-metal corrosion rates of 0.03 mm/y in 30 and 50 wt.% acid, 0.13 mm/y in 70 wt.%, and 0.11 mm/y in 90 wt.%; the corresponding wrought base metal values are even lower. That is not a license to ignore procedure qualification, pickling, or heat tint control, but it is a strong reason C-276 remains a dependable fabricated alloy for sulfuric units.
Quindi, è Hastelloy C-276 corrosion resistance in sulfuric acid good enough for your service? Very often yes—but only after the service is defined properly. Ask for five variables before releasing material: acid concentration, operating and upset temperature, oxidizing contaminants, halides/chlorides, and whether the component is wrought or heavily welded. If you can supply those inputs, material selection becomes engineering instead of guesswork. For serious sulfuric acid duties, that difference is usually what separates a 10-year asset from an expensive shutdown.

Conclusione
In sulfuric acid, C-276 is best understood as a high-margin engineering alloy, not a universal answer. Its Ni-Cr-Mo-W chemistry gives it a broad window, especially where stainless steels fall away, and its welded performance is one of the reasons it is still widely trusted. But the decision becomes truly reliable only when concentration, temperature, and impurity profile are reviewed together against actual process conditions.
Domande e risposte correlate
Q1: Is Hastelloy C-276 better than 316L in sulfuric acid?
In many sulfuric acid conditions, yes. Comparative Haynes plots place C-276 well above 316L in allowable temperature across much of the concentration range, which is why 316L often becomes marginal long before C-276 does.
Q2: Can welded Hastelloy C-276 be used in sulfuric acid service?
Usually yes, provided fabrication is controlled. Haynes data show low corrosion rates for C-276 weld metal in 30 to 90 wt.% sulfuric acid at 66°C, though weld quality, surface condition, and real-stream contaminants still matter.
Q3: What is the biggest mistake when selecting C-276 for sulfuric acid?
Treating sulfuric acid as a single medium. Temperature, oxidizing species, halides, and actual plant impurities can shift corrosion behavior dramatically, so selection should be based on the full liquor chemistry, not nominal H2SO4 concentration alone.


