When metallurgical engineers design critical thermal transfer systems, precise nickel alloy selection for heat exchanger applications becomes the primary defense against catastrophic failure. Processing environments involving aggressive halides, elevated temperatures, and variable flow dynamics rapidly exploit any material weakness. Austenitic stainless steels often succumb to chloride stress corrosion cracking (CSCC) or severe localized pitting under these conditions. Consequently, upgrading to high-performance materials is not merely an option, but an engineering necessity to maintain operational integrity and avoid unplanned facility downtime.
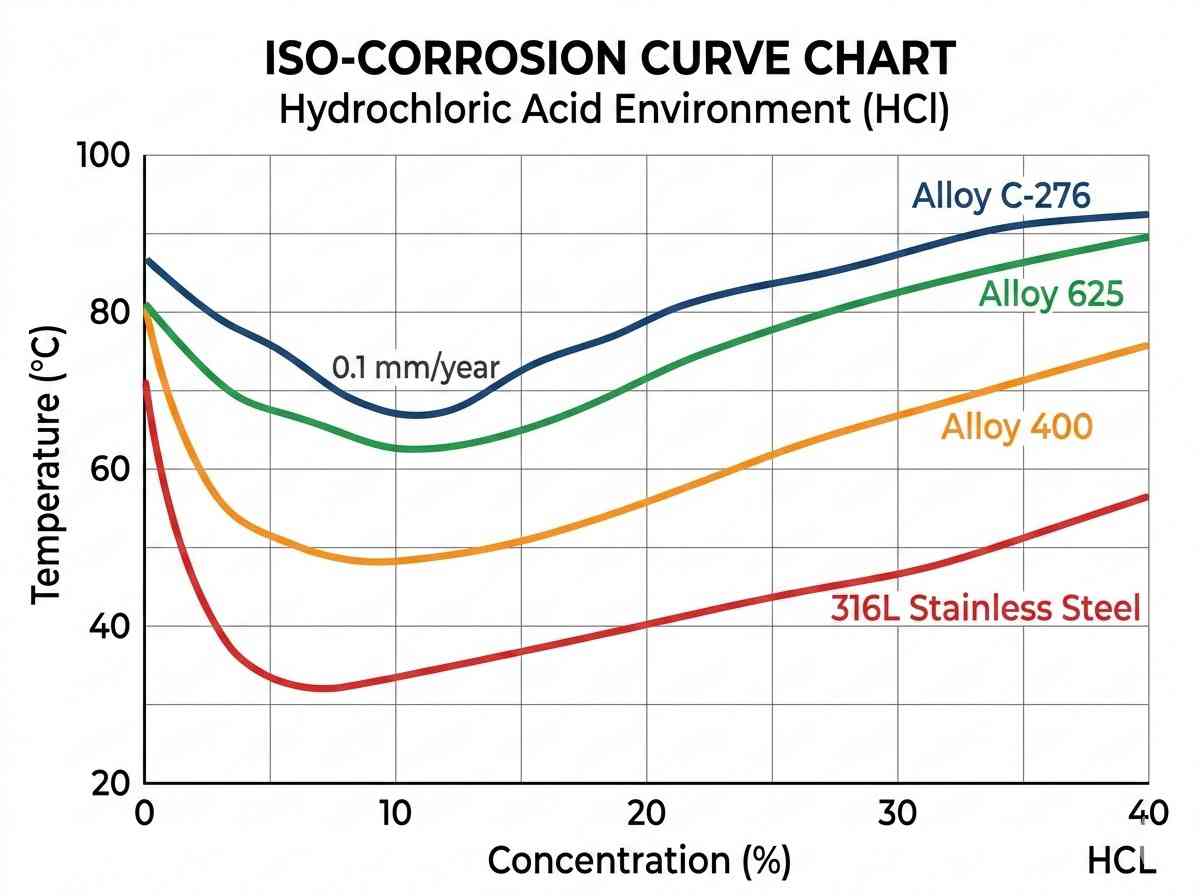
The baseline of nickel alloy selection for heat exchanger design requires a deep understanding of the specific corrosive media present on both the shell and tube sides. Localized corrosion mechanisms, primarily pitting and crevice corrosion, dictate the necessity for high molybdenum and nitrogen additions. When localized chloride concentrations increase—often due to under-deposit corrosion or stagnant flow zones at the tube-to-tubesheet joints—the protective passive oxide layer degrades.

To quantify a material’s resistance to these localized attacks, engineers rely on the Pitting Resistance Equivalent Number (PREN). The formula, generally expressed as %Cr + 3.3(%Mo) + 16(%N), provides a reliable comparative metric. Advanced nickel alloy selection for heat exchanger networks heavily weights this data. For instance, while Alloy 400 performs exceptionally well in hydrofluoric acid environments due to its high copper content, its lack of molybdenum makes it unsuitable for oxidizing chloride environments. Conversely, Alloy C-276, containing roughly 16% molybdenum, offers exceptional resistance to a wide range of aggressive chemicals, including wet chlorine gas and hypochlorite solutions.
Below is a technical comparison of chemical compositions and phase data for common heat exchanger alloys:
| 합금 등급 | 유엔 지정 | Cr (%) | Mo (%) | Ni (%) | 일반적인 PREN | Primary Application Focus |
| 합금 400 | N04400 | – | – | 63.0분 | N/A | Reducing environments, HF acid, marine |
| 합금 600 | N06600 | 14.0-17.0 | – | 72.0분 | ~15 | High-temp oxidation, caustic solutions |
| 합금 625 | N06625 | 20.0-23.0 | 8.0-10.0 | 58.0분 | ~50 | Seawater, high-stress environments |
| 합금 C-276 | N10276 | 14.5-16.5 | 15.0-17.0 | 잔액 | ~68 | Severe oxidizing & reducing chlorides |
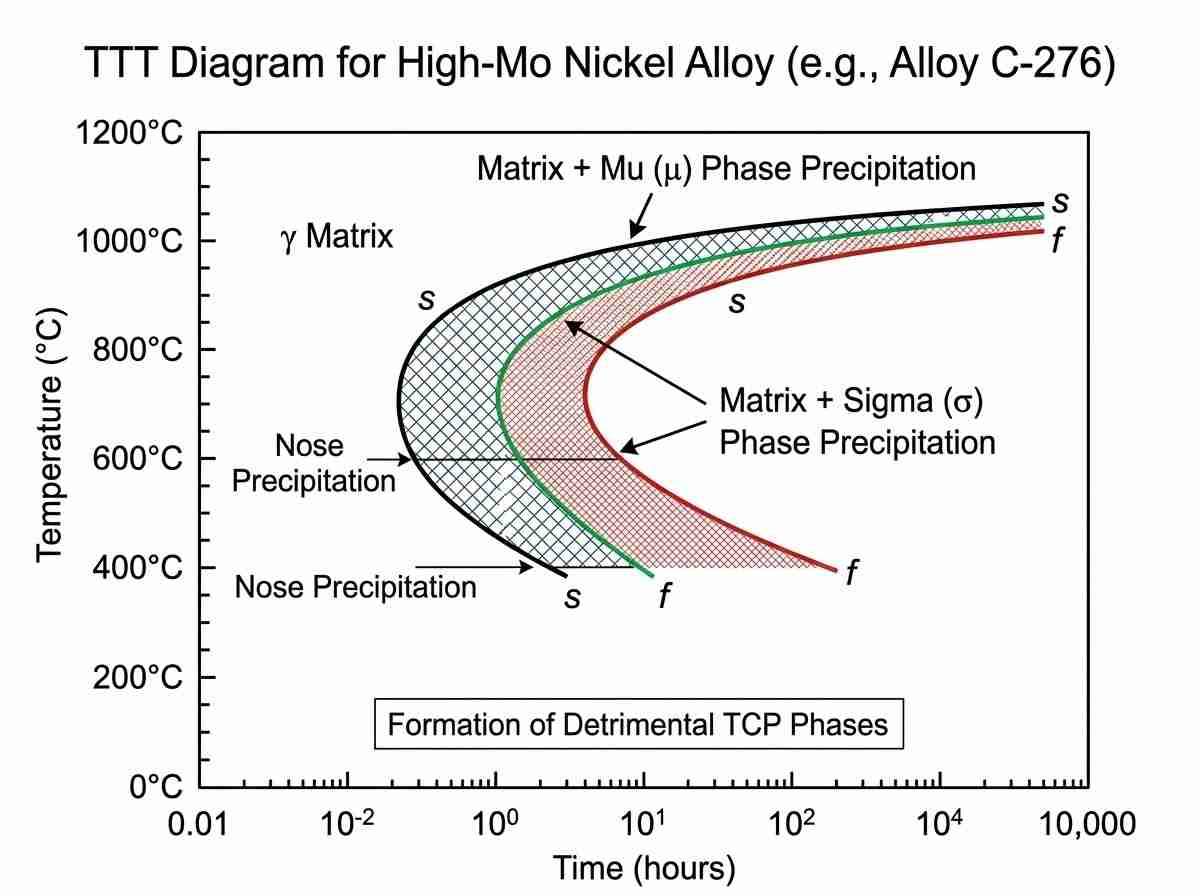
A frequent oversight in nickel alloy selection for heat exchanger applications is the failure to account for phase stability at elevated temperatures, particularly during the fabrication process. While a base metal may possess an ideal PREN, the thermal cycling during welding can induce intermetallic phase precipitation. High-molybdenum grades like Alloy C-276 and Alloy 625 are susceptible to the formation of detrimental topologically close-packed (TCP) phases, such as mu (μ) and sigma (σ) phases, when exposed to temperatures between 650°C and 1000°C.
These intermetallic precipitates severely deplete the adjacent matrix of corrosion-resistant elements, leading to sensitization in the heat-affected zone (HAZ). Furthermore, TCP phases increase localized brittleness, reducing the mechanical integrity of the pressure vessel. Therefore, validating nickel alloy selection for heat exchanger fabrication requires rigorous review of Time-Temperature-Transformation (TTT) curves and specifying appropriate low-heat-input welding procedures to suppress precipitation kinetics.
Beyond corrosion parameters, structural mechanics play a vital role. The coefficient of thermal expansion (CTE) must be carefully matched if the shell and tubes are constructed from dissimilar metals. Thermal fatigue, driven by cyclic expansion and contraction, can induce stress concentrations at the baffles and tube-to-tubesheet welds. Selecting a nickel alloy with a CTE compatible with the carbon steel or duplex stainless steel shell minimizes these induced shear stresses, thereby extending the mechanical fatigue life of the entire thermal unit.
Ultimately, mastering nickel alloy selection for heat exchanger longevity is not a guesswork exercise; it demands rigorous analysis of localized corrosion data, phase stability kinetics, and mechanical thermodynamics. Off-the-shelf solutions rarely apply to complex chemical processing units. Our engineers at 28Nickel possess the deep metallurgical expertise required to analyze your specific operational parameters and assist in specifying the exact microstructural requirements for your equipment. Contact our technical team today to share your process data and receive dedicated engineering support.
관련 Q&A
1. Is chloride concentration the only factor in nickel alloy selection for heat exchanger units? No. While chlorides drive pitting and CSCC, temperature, pH, fluid velocity, and the presence of oxidizing agents (like ferric or cupric ions) drastically alter the corrosion profile. An environment with low chlorides but high oxidizers often requires a higher chromium-to-molybdenum ratio than a purely reducing chloride environment.
2. Why does welding impact nickel alloy selection for heat exchanger fabrication? Welding introduces intense thermal gradients. Alloys with high refractory metal content (like Mo and W) can precipitate intermetallic phases in the heat-affected zone (HAZ) during welding. This sensitization lowers local corrosion resistance and impacts mechanical ductility, dictating the need for specific filler metals and controlled heat inputs.
3. How does fluid velocity dictate the use of Alloy C-276 versus Alloy 625? Stagnant or low-velocity fluids promote under-deposit corrosion and severe crevice attack, strongly favoring Alloy C-276 due to its superior localized corrosion resistance (higher PREN). In higher velocity conditions where erosion-corrosion is the primary threat, the higher yield strength and work-hardening characteristics of Alloy 625 may provide a mechanical advantage.