When engineers ask about Monel 400 corrosion resistance in sulfuric acid, my first response is usually the same: do not treat it as a simple yes-or-no materials question. MONEL alloy 400 is a nickel-copper, solid-solution alloy, typically with at least 63% nickel and 28–34% copper, and that metallurgy gives it a very specific corrosion profile. It performs well in many reducing environments, but sulfuric acid is only “friendly” to Monel 400 when the chemistry stays on that side of the line. Once oxygen, oxidizing salts, concentration shifts, or temperature push the acid into a more oxidizing regime, the answer changes quickly.
That distinction matters in real plants. People sometimes specify Monel 400 because they know it handles hydrofluoric acid, seawater, or non-oxidizing chlorides extremely well. Then the same alloy gets carried into sulfuric acid service by analogy. That is where mistakes begin. In sulfuric acid, alloy selection is not governed by the nameplate acid alone; it is governed by concentration, temperature, aeration, contaminants, flow pattern, and whether corrosion products are allowed to accumulate in the loop.
When Monel 400 Corrosion Resistance in Sulfuric Acid Is Strong
Sulfuric acid is generally reducing up to roughly 25% concentration, but as concentration rises it begins to take on oxidizing characteristics; commercially concentrated acid above about 87 wt% at room temperature is commonly oxidizing in nature. That is the first principle behind Monel 400 corrosion resistance in sulfuric acid. The alloy is at its best when the acid remains reducing. Special Metals states plainly that MONEL alloy 400 is used for sulfuric acid solutions under reducing conditions, and also notes that it is resistant to many forms of sulfuric acid under reducing conditions.
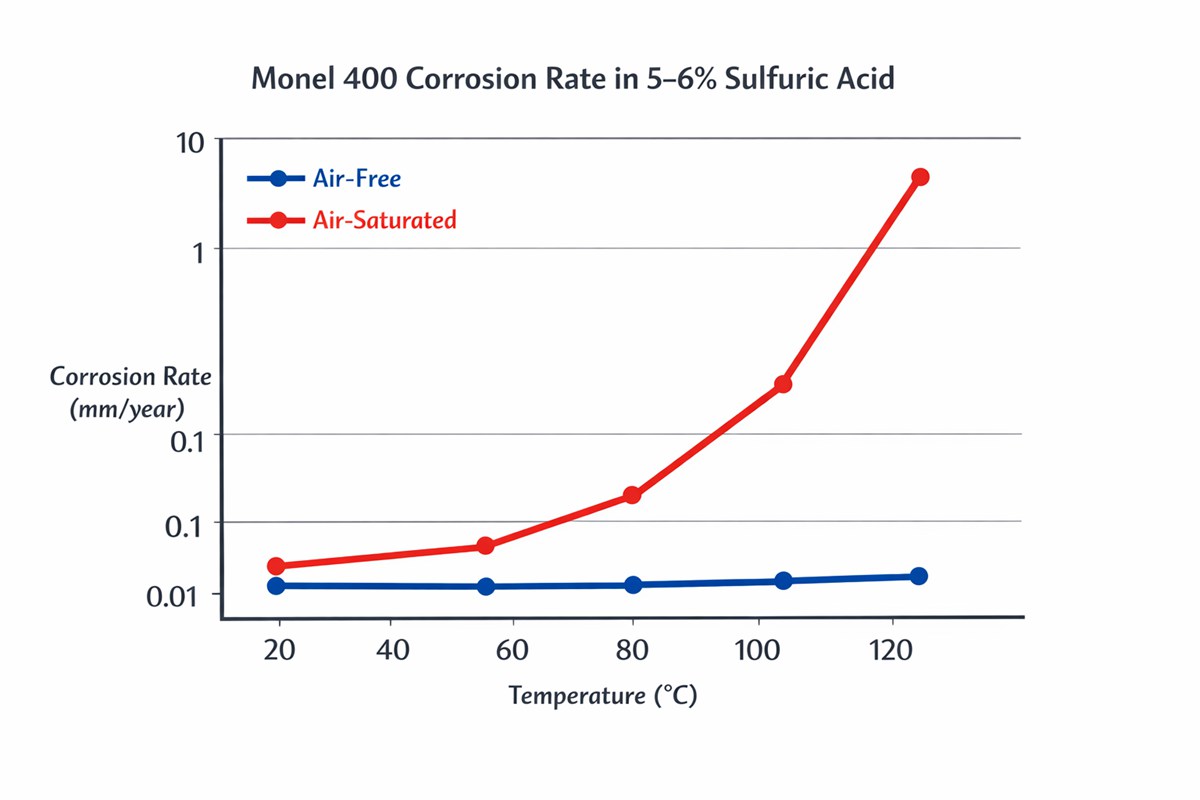
The second principle is aeration. In 5–6% sulfuric acid, the published Special Metals data show very low corrosion rates in air-free solution over the tested temperature range. In air-saturated acid, however, the corrosion rate rises sharply with temperature, peaks around the higher end of the sub-boiling range, and only drops back down near the boiling point, where it approaches the air-free case again. That is not a subtle effect. For designers, it means a tank, loop, or absorber that picks up oxygen can behave very differently from a quiet, closed, reducing system, even at the same nominal acid concentration.
Special Metals also reports that Monel 400 has shown suitable resistance in boiling sulfuric acid solutions up to about 15% concentration. In practice, it has also shown satisfactory resistance for storage of 80% acid at room temperature. Rolled Alloys echoes that field experience, noting that Alloy 400 is often chosen for sulfuric acid through 80% concentration at room temperature, and for about 15% sulfuric acid at boiling. Those are useful guideposts, but they are not blanket approvals for every process line.
Why Monel 400 Corrosion Resistance in Sulfuric Acid Can Collapse
The trap is assuming the alloy remains stable just because the acid concentration looks acceptable on paper. In reality, oxidizing salts can make sulfuric acid far more aggressive to Monel 400. Special Metals specifically warns about ferric sulfate, chromates, dichromates, nitrates, nitrites, peroxides, and cupric salts. In recirculating or long-residence systems, intermediate sulfuric acid concentrations can also become autocatalytically more corrosive because cupric ions build up in solution from ongoing attack of the nickel-copper alloy itself. That is the kind of mechanism that does not show up on a simple purchasing spec sheet, but it absolutely shows up in premature field failures.
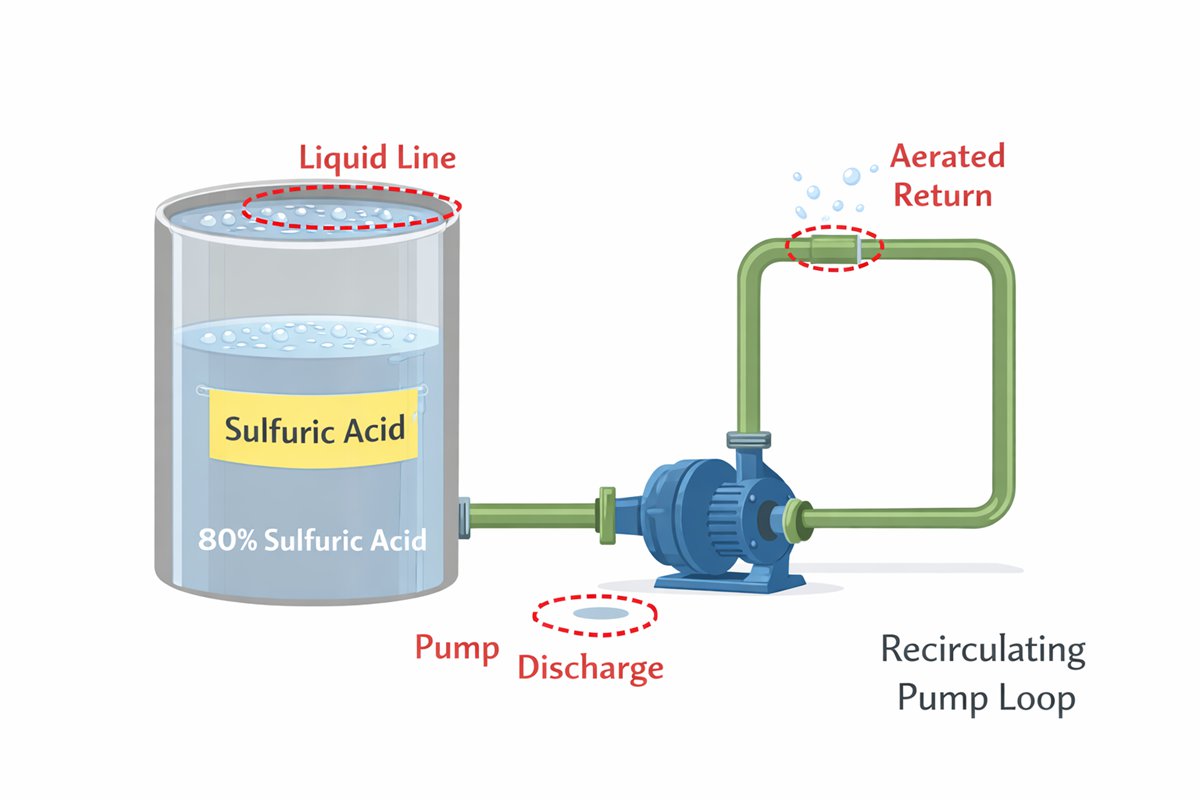
Velocity matters too. The usual effect of increasing metal-to-liquid relative motion is to raise corrosion rate because fresh acid and dissolved oxygen are brought continuously to the surface while the diffusion layer is thinned. For Monel 400, the velocity effect is most pronounced in aerated sulfuric acid. So if your service involves spraying, splashing, entrained air, pump recirculation, or a persistent liquid line, you are no longer evaluating a calm reducing-acid condition. You are evaluating an oxygen-renewed corrosion cell.
One more point that engineers sometimes overlook: Special Metals states that boiling 25% sulfuric acid attacks Monel 400, even though the alloy may perform acceptably in boiling solutions up to about 15%. That gap tells you something important. The alloy’s service window is real, but it is not broad enough to justify “close enough” decision-making. In sulfuric acid service, a seemingly small change in concentration or operating temperature can move Monel 400 from dependable to risky.
The table below is a practical screening summary synthesized from Special Metals and Rolled Alloys service guidance. It is useful for early material selection, but it should never replace plant-specific coupon testing or a full process-chemistry review.
| Sulfuric acid condition | Expected Monel 400 behavior | Engineering interpretation |
|---|---|---|
| 5–6% H₂SO₄, air-free | Very low corrosion rate across tested temperatures | Strong candidate when the system stays truly reducing |
| 5–6% H₂SO₄, air-saturated | Corrosion rate rises strongly with temperature before falling near boiling | Oxygen ingress can dominate the material decision |
| Boiling sulfuric acid up to about 15% | Suitable resistance reported | Reasonable service window, but verify actual velocity and impurities |
| Boiling 25% sulfuric acid | Active attack reported | Not a comfortable Monel 400 duty point |
| 80% sulfuric acid at room temperature, storage duty | Satisfactory resistance reported in practice | Storage service is not the same as hot, aerated, recirculating service |
| Pure sulfuric acid above ~85% or at elevated temperature | Oxidizing behavior increases risk sharply | Require validation testing; alternative alloys may be safer |
What follows from that table is straightforward engineering logic. If your sulfuric acid service is closed, reducing, relatively clean, and not strongly aerated, Monel 400 corrosion resistance in sulfuric acid can be very attractive. If the service is hot, oxygen-rich, contaminated with oxidizers, or subject to long recirculation at intermediate concentrations, the alloy moves out of its comfort zone fast.
What Experienced Materials Engineers Check Before Approving the Alloy
In real projects, I would not sign off on Monel 400 for sulfuric acid service until five questions were answered clearly: what is the true acid concentration at the metal surface, not just in the bulk stream; what is the dissolved oxygen level; are ferric or cupric species present; is the service static storage or recirculating process duty; and what upset conditions can temporarily shift the chemistry into a more oxidizing range. That is where many “datasheet-safe” selections fail in operation. The handbook itself notes that actual corrosion rates are often closer to the air-free case because continuous air saturation is uncommon, but it also warns that corrosion can accelerate at the liquid line and in situations with strong aeration.
There is also a useful selection boundary here. Special Metals notes that INCONEL alloy 600, because of its chromium content, offers better sulfuric acid resistance under oxidizing conditions than Nickel 200 or Monel 400, while more aggressive hot sulfuric acid environments are often handled by higher-alloy nickel-chromium-molybdenum grades such as 625, 622, C-276, or 686. In other words, Monel 400 is not a universal sulfuric-acid alloy. It is a very good reducing-acid alloy within a defined envelope. That is an important distinction for both engineers and procurement teams trying to balance corrosion margin against cost.
Special Metals also reports that Monel 400 is generally not susceptible to stress-corrosion cracking in sulfuric acid, except in solutions containing mercury salts or considerable hydrofluoric or fluorosilicic acid; in such cases, stress-relief heat treatment before service is recommended. That detail matters in fabricated equipment, especially where residual welding stress and mixed-acid contamination are realistic.
Conclusion
So, is Monel 400 a good sulfuric-acid material? Yes—but only when you respect the chemistry. Monel 400 corrosion resistance in sulfuric acid is strongest in reducing, relatively low-oxygen service, and it remains useful in some boiling dilute-acid and room-temperature concentrated-acid storage scenarios. But once sulfuric acid becomes more oxidizing, aerated, contaminated, or aggressively recirculated, the alloy can lose its margin much faster than many buyers expect. That is why serious material selection should be based on the real process envelope, not the nominal acid name alone.
If your team is evaluating plate, pipe, fittings, flanges, or fasteners for sulfuric acid duty, the most practical next step is a service-window review built around concentration, temperature, oxygen pickup, impurities, and expected upset conditions. That is usually where the right alloy choice becomes obvious.
Related Q&A
Q1: Can Monel 400 be used in concentrated sulfuric acid?
Yes, but only in a limited and conditional sense. Published guidance says Monel 400 has shown satisfactory resistance in 80% sulfuric acid at room temperature for storage duty, while pure acid at higher concentration should not be used continuously without preliminary testing. Special Metals also notes that above about 85%, sulfuric acid becomes oxidizing and corrosion risk rises sharply.
Q2: Why does dissolved air matter so much for Monel 400 in sulfuric acid?
Because Monel 400 is fundamentally more comfortable in reducing sulfuric acid than in oxidizing sulfuric acid. Special Metals’ published data for 5–6% acid show that air-free conditions give very low corrosion rates, whereas aeration can increase attack significantly. In practice, dissolved oxygen, splashing, bubbling, and pump recirculation can shift a seemingly mild acid service into a much harsher one.
Q3: Is Monel 400 better than chromium-bearing nickel alloys in sulfuric acid?
Not across the board. Under reducing sulfuric acid conditions, Monel 400 can be excellent. Under oxidizing conditions, chromium-bearing alloys can be better. Special Metals specifically states that INCONEL alloy 600 offers better resistance than Monel 400 in sulfuric acid under oxidizing conditions, and that more aggressive hot sulfuric acid environments are often handled by higher-alloy Ni-Cr-Mo grades.