Material degradation in aggressive process environments rarely announces itself before a critical failure occurs. For engineers designing systems in petrochemical, desalination, or power generation sectors, specifying the correct alloy is a high-stakes decision. The core debate often centers on a specific metallurgical threshold: evaluating nickel alloy vs stainless steel for boundary-condition applications. While both material families rely on a passive oxide layer for corrosion resistance, their base matrices dictate fundamentally different responses to thermal fatigue, chloride-induced stress, and highly acidic process fluids. Understanding the distinct metallurgical mechanisms behind these materials is essential for preventing catastrophic localized failures and optimizing the lifecycle of critical infrastructure.

Metallurgical Baselines: Iron vs. Nickel Matrices
To properly evaluate nickel alloy versus stainless steel performance, we must first look at the solvent element of the metallurgical matrix. Stainless steels are iron-based alloys containing a minimum of 10.5% chromium. Standard austenitic grades, like 316L, incorporate around 10-14% nickel and 2-3% molybdenum to stabilize the austenitic structure and enhance localized corrosion resistance. However, the iron-dominated lattice remains susceptible to specific failure modes under extreme duress.
Conversely, nickel alloys (such as Inconel®, Hastelloy®, or Monel®) utilize nickel as the primary base metal rather than iron. This shift in the baseline matrix drastically alters the material’s thermodynamic stability. Nickel readily accommodates high weight percentages of alloying elements like chromium, molybdenum, and tungsten in solid solution without forming detrimental intermetallic phases (such as Sigma phase) that often plague highly alloyed stainless steels during thermal cycling.
Quantitative Performance Comparison
The following table highlights the threshold differences between a premium austenitic stainless steel and a standard solid-solution strengthened nickel alloy:
| Metallurgical Parameter | 316L Austenitic Stainless Steel | Alloy 625 (Nickel Alloy) |
| Primary Base Element | Iron (Fe) | Nickel (Ni) |
| Nominal Nickel Content | 10.0% – 14.0% | 58.0% Minimum |
| Molybdenum Content | 2.0% – 3.0% | 8.0% – 10.0% |
| Typical Yield Strength | ~170 MPa | ~414 MPa |
| PREN (Pitting Resistance) | ~24 | ~50 |
| Chloride SCC Resistance | Low (Susceptible >60°C) | Highly Immune |
| Max Service Temperature | ~870°C (Scaling occurs) | ~980°C (High oxidation resistance) |
The Chloride Threshold and High-Temperature Creep
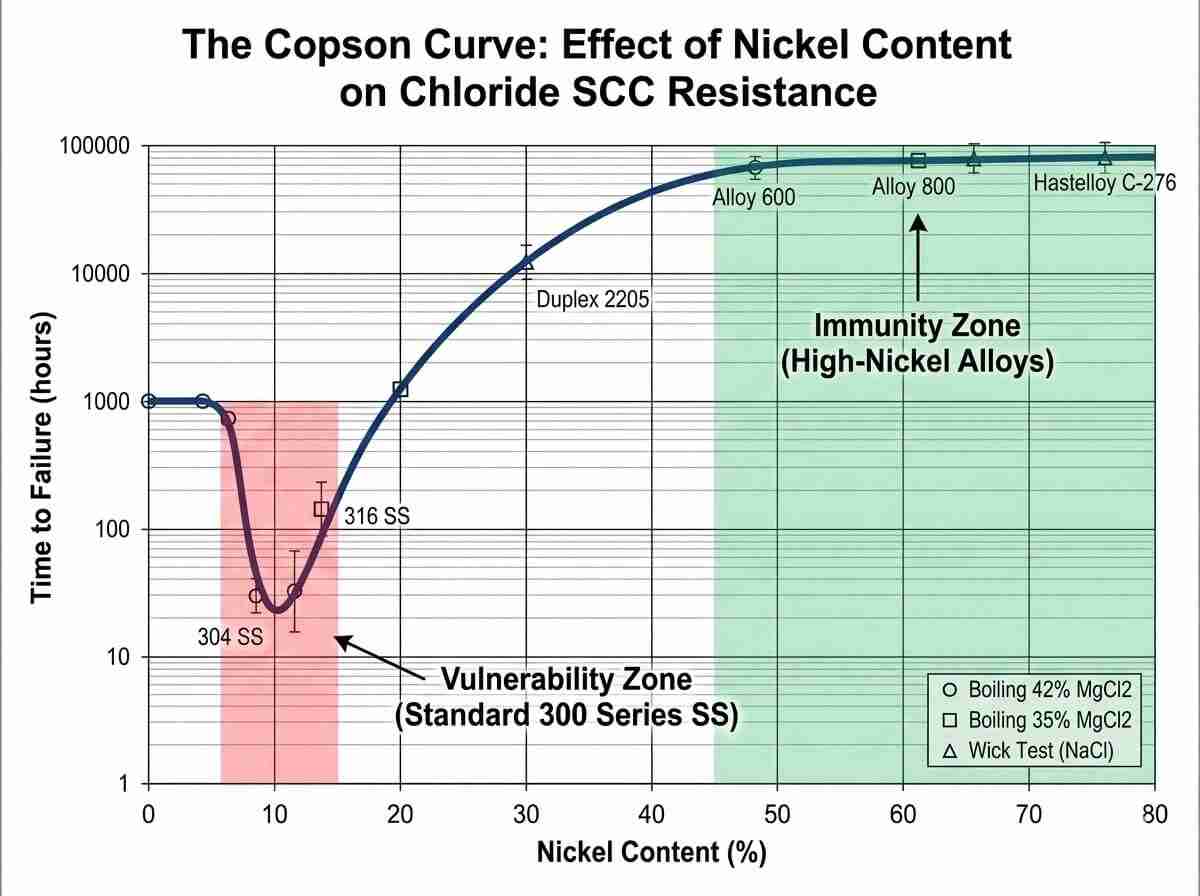
One of the most defining factors in the stainless steel versus nickel alloys selection process is the phenomenon of Chloride Stress Corrosion Cracking (CSCC). Austenitic stainless steels are notoriously vulnerable to CSCC. According to the Copson curve, materials with a nickel content between 8% and 12%—which happens to be the exact range of standard 300-series stainless steels—exhibit the highest susceptibility to rapid cracking in warm chloride environments. When process temperatures exceed 60°C in the presence of tensile stress and chlorides, an iron-based lattice can fail in a matter of days. High-nickel alloys completely bypass this vulnerability. By pushing the nickel mass fraction above 42%, the atomic lattice becomes virtually immune to chloride-induced transgranular cracking.
Furthermore, engineers must calculate the Pitting Resistance Equivalent Number (PREN) to predict performance in sour gas or high-chloride acidic conditions. The governing formula is: PREN=%Cr+3.3×%Mo+16×%N While high-end super duplex stainless steels might reach a PREN of 40, nickel-molybdenum-chromium alloys routinely exceed a PREN of 50, providing an impenetrable barrier against localized pitting.
Beyond corrosion, thermal creep dictates material choice in gas turbines and reactor vessels. Nickel alloys maintain structural integrity at elevated temperatures where iron-based alloys rapidly lose tensile strength. The face-centered cubic (FCC) structure of nickel alloys allows for the precipitation of gamma prime phase (γ′), offering exceptional creep-rupture strength at temperatures pushing 1000°C.
Engineering Validation
Specifying the correct metallurgical boundary requires analyzing your specific media concentration, operating temperature, and stress loads. Over-engineering with a high-nickel alloy increases capital expenditure, while under-engineering with a standard stainless steel guarantees disastrous operational downtime. If your current operational parameters are pushing your metallurgy to the absolute limit, reach out to the engineering team at 28Nickel. We can evaluate your exact chemical environments and thermal cycling data to provide a rigorously calculated material recommendation for your next critical fabrication.
Related Q&A
Q: Can stainless steel be welded to a high-nickel alloy in a pressure vessel system?
A: Yes, dissimilar metal welding is common, but it requires precise filler metal selection to prevent galvanic corrosion and thermal expansion mismatches. Generally, a nickel-based filler metal, such as ERNiCrMo-3 (Alloy 625), is utilized because it can absorb dilution from the iron-based stainless steel without forming crack-sensitive microstructures in the weld pool.
Q: Is Duplex stainless steel a viable middle-ground alternative to nickel alloys?
A: Duplex stainless steels (ferritic-austenitic) offer excellent chloride stress corrosion cracking resistance and higher yield strength than 300-series stainless steels, often serving as a cost-effective bridge. However, they are metallurgically limited by an upper service temperature threshold of approximately 250°C. Above this, they suffer from “475°C embrittlement,” making nickel alloys the mandatory choice for high-temperature applications.
Q: How does the presence of Molybdenum dictate the choice between these two metal families?
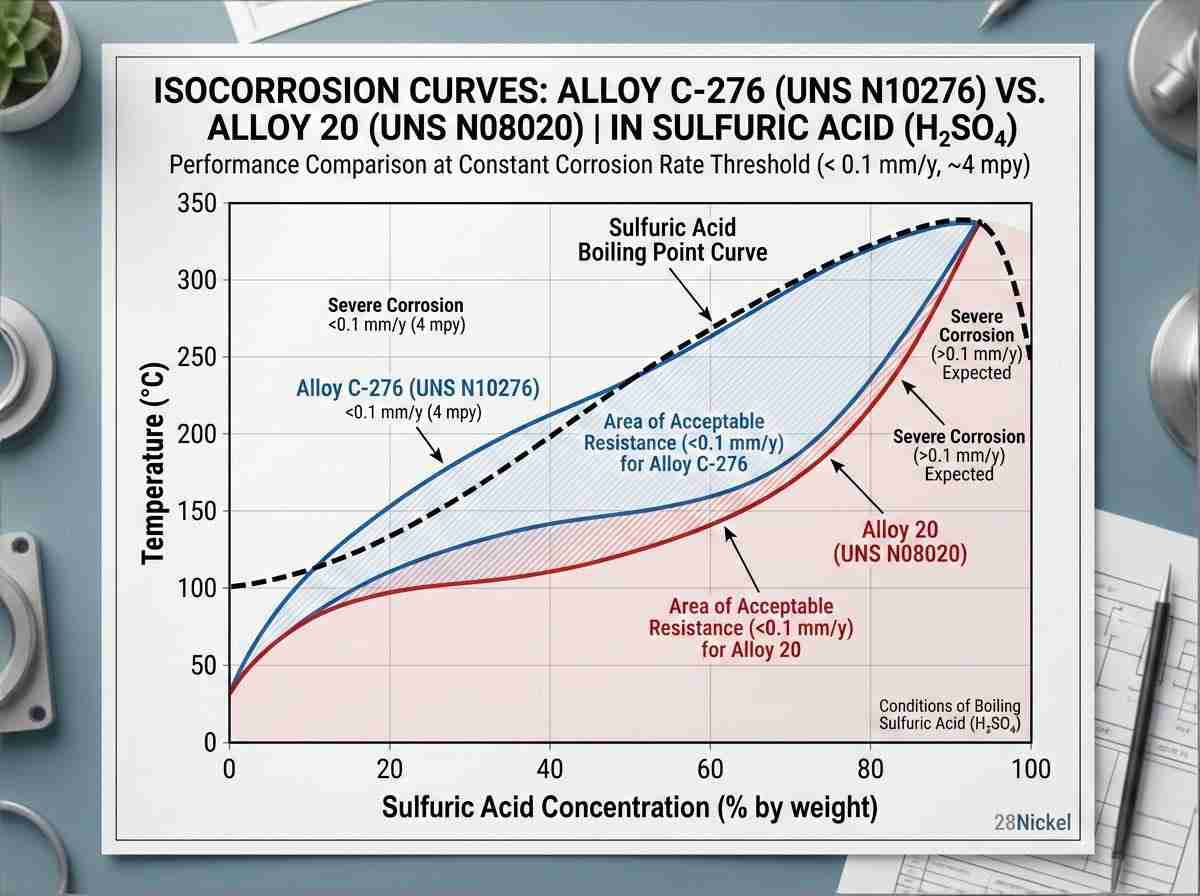
A: Molybdenum is the primary driver for resisting localized pitting and crevice corrosion in reducing acid environments (like hydrochloric or sulfuric acid). While 316L stainless steel maxes out around 3% Mo, advanced nickel alloys like Hastelloy C-276 contain up to 16% Mo. If your fluid analysis shows active reducing chlorides, the higher molybdenum solubility limit of the nickel matrix is required to prevent rapid localized wall loss.