In the realm of advanced material science and high-performance engineering, few material classes hold the prominence and versatility of nickel alloys. Far beyond pure nickel, nickel alloys are sophisticated metallic composites designed to excel under extreme conditions—temperatures, pressures, and corrosive environments—where common steels and other metals would fail. Understanding what are nickel alloys and their unique metallurgical properties is crucial for industries from aerospace to chemical processing.
The Metallurgy Behind Nickel Alloys: Composition and Structure
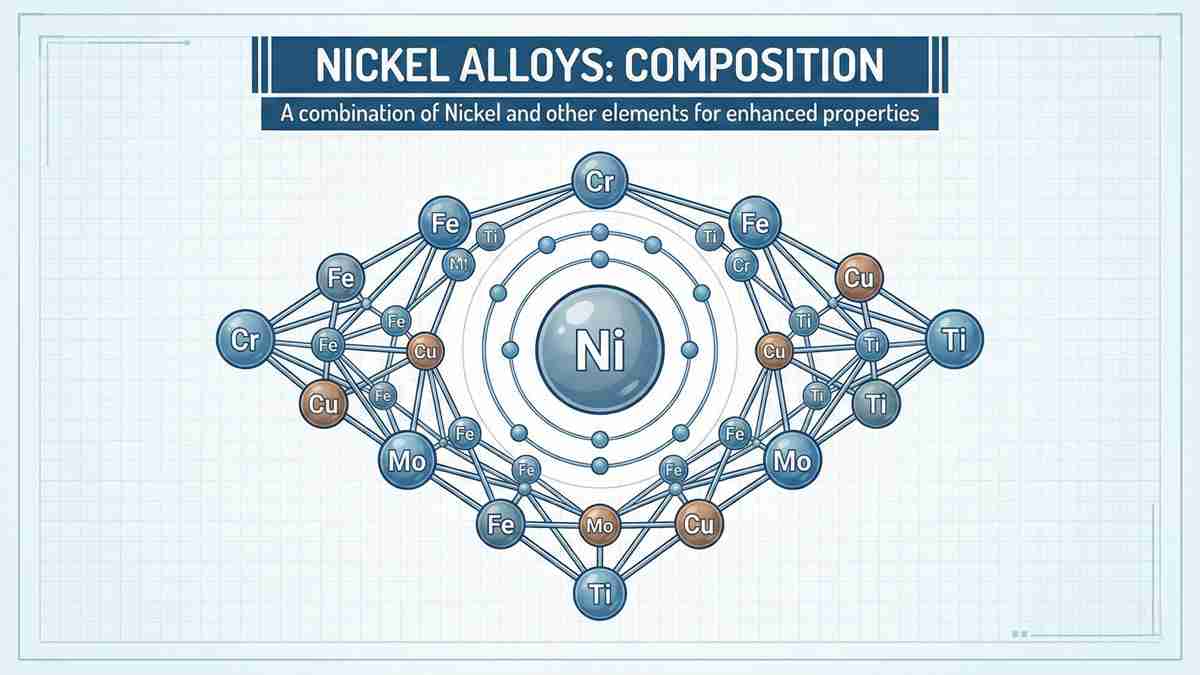
Defining what are nickel alloys starts with their base metal: nickel. A typical nickel alloy contains nickel as its primary constituent, often exceeding 50% by weight. The exceptional properties of nickel alloys are achieved by strategically introducing alloying elements such as chromium (Cr), iron (Fe), molybdenum (Mo), and copper (Cu).
-
Chromium: Provides superior oxidation resistance and high-temperature strength, essential for superalloys.
-
Molybdenum: Significantly enhances resistance to localized corrosion, particularly pitting and crevice corrosion, making it key in certain nickel alloys like Hastelloy.
-
Iron: Often used to balance cost while maintaining necessary mechanical strength in specific nickel alloys (e.g., Incoloy series).
The microstructure of nickel alloys is often characterized by a face-centered cubic (FCC) crystal structure, which contributes to their excellent ductility and resistance to fracture, even at cryogenic temperatures. This stable structure is fundamental to the reliable performance of these specialized nickel alloys.
Key Advantages: Why Choose Nickel Alloys?
The engineering advantages of nickel alloys are directly linked to their complex compositions. Engineers frequently select nickel alloys over stainless steels or titanium for applications demanding a confluence of the following properties:
-
Exceptional Corrosion Resistance: Many nickel alloys (e.g., Monel and Inconel) form a tenacious, self-healing, passive oxide layer—typically chromia (Cr2O3)—which provides unparalleled resistance to various aggressive media, including acids, alkalis, and chlorides. This is the core answer to the question of what are nickel alloys best used for.
-
Outstanding High-Temperature Strength (Creep Resistance): Superalloys, a subset of nickel alloys (like Waspaloy and René alloys), maintain their structural integrity and strength at temperatures exceeding 1000∘C (1832∘F), making them indispensable for jet engine components.
-
High Ductility and Toughness: The inherent FCC structure gives nickel alloys the ability to absorb significant energy before fracture, making them reliable in high-stress and impact scenarios.
-
Magnetostriction and Thermal Expansion Control: Specific nickel alloys (like Kovar and Invar) are designed for controlled thermal expansion, critical for precision electronic and optical applications.
Critical Applications of Nickel Alloys
The superior performance profile of nickel alloys has cemented their role across numerous high-stakes industries:
| Industry | Primary Use of Nickel Alloys | Key Property Utilized |
| Aerospace | Turbine blades, combustors, exhaust systems | High-temperature creep and oxidation resistance |
| Chemical Processing | Reactor vessels, heat exchangers, piping | Resistance to hot, aggressive acids and chlorides |
| Oil & Gas | Downhole equipment, subsea components | Stress corrosion cracking (SCC) resistance |
| Power Generation | Nuclear reactor control rod mechanisms | Neutron absorption and long-term stability |




