Material degradation in the petrochemical, offshore, and chemical processing sectors costs billions annually. When critical infrastructure faces aggressive chloride environments, acidic streams, or elevated temperatures, standard austenitic stainless steels fail rapidly through pitting, crevice corrosion, or stress corrosion cracking (SCC). In these high-stakes scenarios, engineers inevitably ask: what exactly is the best nickel alloy for corrosion resistance?
The reality of materials engineering is that a universal, invincible alloy does not exist. Material selection is an exercise in balancing localized chemistry, operating temperatures, and structural stability. To find the optimal solution, we must analyze the metallurgical mechanisms that govern passivity and active dissolution.

Assessing High-Performance Grades via PREN
When evaluating the best nickel alloy for corrosion resistance, metallurgists often begin with the Pitting Resistance Equivalent Number (PREN). This predictive index quantifies a metal’s resistance to localized pitting in chloride-bearing environments based on its chemical composition. For Ni-Cr-Mo alloys, the standard formula often incorporates tungsten (W) due to its synergistic effect with molybdenum:
PREN=%Cr+3.3(%Mo+0.5%W)+16(%N)
Alloy C-276 (UNS N10276) has long been considered the industry workhorse, boasting excellent resistance to localized attack. However, as process conditions have become more severe, newer alloys have been engineered to push the boundaries of passivity. Alloy 59 (UNS N06059), for instance, achieves a significantly higher PREN by maximizing chromium and molybdenum content while virtually eliminating tungsten and lowering iron. Therefore, if your primary failure mode is chloride-induced pitting, determining the best nickel alloy for corrosion resistance requires looking closely at these precise elemental ratios to understand breakdown potential.
| Alloy Grade | UNS Designation | Chromium (%) | Molybdenum (%) | Tungsten (%) | PREN (approx.) |
| Alloy 625 | N06625 | 20.0 – 23.0 | 8.0 – 10.0 | – | 45 – 50 |
| Alloy C-276 | N10276 | 14.5 – 16.5 | 15.0 – 17.0 | 3.0 – 4.5 | ~68 |
| Alloy 22 | N06022 | 20.0 – 22.5 | 12.5 – 14.5 | 2.5 – 3.5 | ~74 |
| Alloy 59 | N06059 | 22.0 – 24.0 | 15.0 – 16.5 | – | >76 |
Oxidizing vs. Reducing Process Environments
Relying solely on PREN data is a dangerous oversimplification. The best nickel alloy for corrosion resistance in one acid stream might degrade rapidly in another due to the fundamental difference between oxidizing and reducing environments.
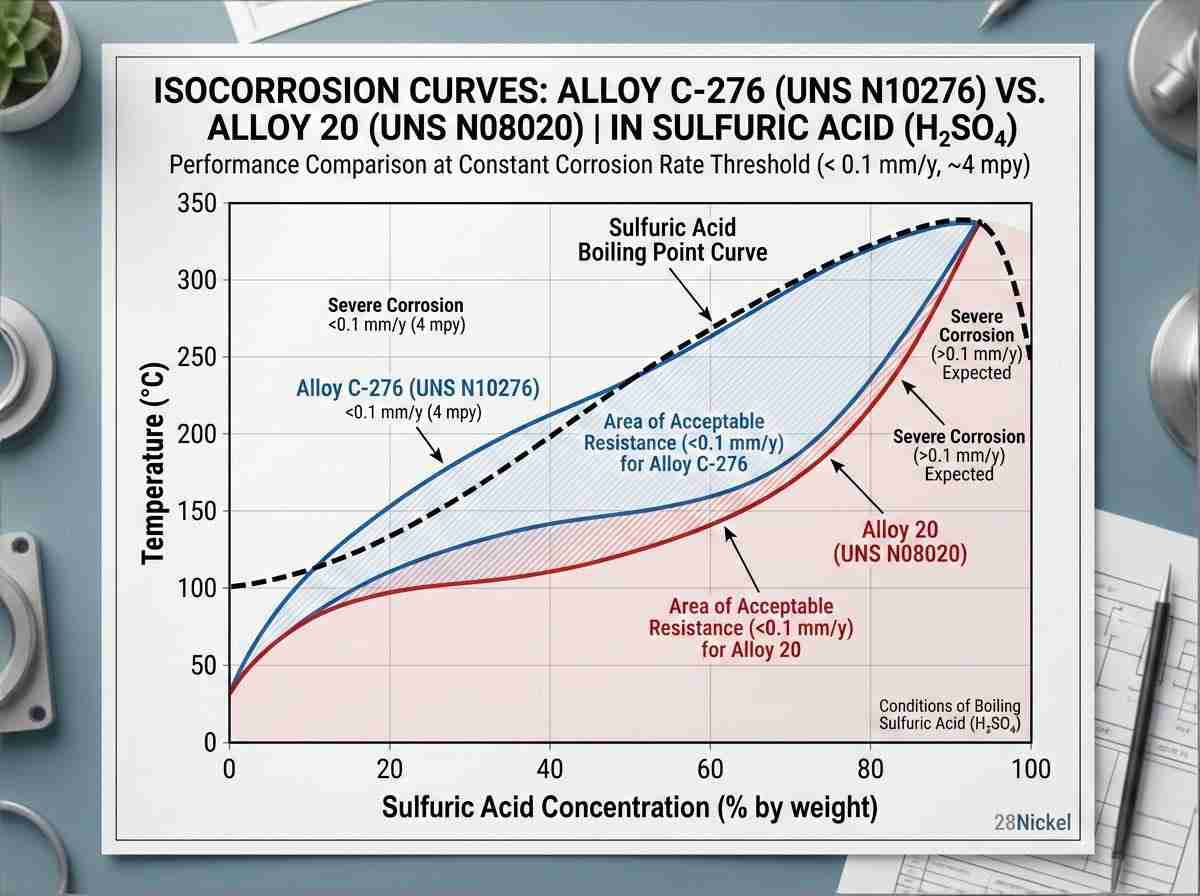
In reducing acids, such as pure hydrochloric (HCl) or dilute sulfuric acid (H2SO4), anodic dissolution is the primary threat. Here, high molybdenum content is the critical defense mechanism, slowing down active dissolution kinetics. Alloy C-276 and Alloy B-3 excel in these specific conditions.
Conversely, in strong oxidizing media like wet chlorine gas, nitric acid (HNO3), or streams containing ferric/cupric ions, the metal relies on chromium to rapidly form a stable, impermeable oxide layer. Alloy C-276, with its relatively lower chromium content (approx. 16%), can suffer in severe oxidizing conditions. In such cases, Alloy 22 or Alloy 59 (both exceeding 20% Cr) become the superior choices. Furthermore, when the process stream fluctuates between reducing and oxidizing states, identifying the best nickel alloy for corrosion resistance becomes highly complex. Alloy C-2000 (UNS N06200) was specifically engineered for this dilemma; the strategic addition of 1.6% copper enhances its resistance to reducing acids while maintaining high chromium for oxidizing conditions.

Microstructural Stability and Thermal Sensitization
Bulk chemical composition dictates theoretical performance, but fabrication dictates practical reality. A frequently overlooked factor in determining the best nickel alloy for corrosion resistance is microstructural stability during thermal cycling and welding.
When heavy-wall sections are welded, the heat-affected zone (HAZ) experiences slow cooling rates. In alloys heavily alloyed with tungsten and molybdenum (like C-276), this thermal exposure can trigger the precipitation of detrimental intermetallic phases (such as the μ-phase) and grain boundary carbides. These precipitates deplete the surrounding matrix of corrosion-resistant elements, leading to severe intergranular corrosion (IGC) in service.
Modern iterations, such as Alloy 59 and Alloy 22, feature ultra-low carbon and silicon limits, coupled with balanced solid-solution strengtheners, to dramatically improve thermal stability. Consequently, the best nickel alloy for corrosion resistance in a complex, multi-pass welded pressure vessel may differ entirely from the alloy chosen for a seamless, straight-run tubing system.
Reaching an Engineering Consensus
Ultimately, selecting the best nickel alloy for corrosion resistance is not about finding the most expensive grade on the market; it is about matching the metallurgical profile to the exact thermodynamic and chemical realities of your process fluid. A shift of 10°C, a slight drop in pH, or a spike in trace chlorides can completely alter the localized degradation mechanism.
At 28Nickel, our metallurgy team relies on empirical testing data, potentiodynamic polarization curves, and deep failure analysis to resolve these exact engineering puzzles. If you are struggling with unexpected material degradation or designing equipment for a new, aggressive process stream, share your specific environment parameters—temperature, pH, chloride concentration, and redox potential—with our engineering desk. We will provide a rigorous metallurgical assessment to help you specify the exact grade your infrastructure requires.
Related Q&A
Q: Does a higher molybdenum content always guarantee better corrosion resistance? A: Not necessarily. While molybdenum is crucial for resisting reducing acids and localized pitting, excessively high molybdenum without a balance of chromium can compromise the alloy’s stability in highly oxidizing environments. Furthermore, over-alloying with Mo can reduce thermal stability, leading to detrimental intermetallic phase precipitation during welding.
Q: Can Alloy 625 safely replace Alloy C-276 in sour gas applications? A: It depends strictly on the hydrogen sulfide (H2S) concentration, temperature, and partial pressure of chlorides. Alloy 625 performs well in mildly sour environments, but under severe sour gas conditions with high temperatures, Alloy 625 is highly susceptible to SCC. Alloy C-276 or Alloy 718 (in precipitation-hardened states) are typically required for extreme downhole sour environments.
Q: How does the addition of copper affect the performance of Alloy C-2000? A: The deliberate addition of approximately 1.6% copper to the Ni-Cr-Mo matrix of Alloy C-2000 significantly widens its operational window. Copper substantially decreases the corrosion rate in reducing acids (like sulfuric and hydrofluoric acids) by altering the cathodic reaction kinetics, while its high chromium content simultaneously maintains excellent resistance to oxidizing media.