Engineers designing components for offshore platforms, hydrofluoric acid alkylation units, or seawater desalination plants face a relentless thermodynamic reality: localized halide corrosion. When standard austenitic stainless steels succumb to chloride stress corrosion cracking (CSCC), specifying a more robust metallurgical matrix becomes non-negotiable. This is precisely where nickel alloy monel demonstrates its structural and chemical superiority. By relying on a solid-solution binary system rather than easily compromised chromium passivation layers, this material mitigates the initiation of pitting in aggressive electrolytes. For engineers at 28Nickel analyzing failure modes, understanding this specific alloy system is critical.
The Metallurgical Architecture of Nickel Alloy Monel
The fundamental advantage of this material lies in the complete mutual solubility of nickel and copper. Because these two elements have similar atomic radii and electronegativity, they form a single-phase face-centered cubic (FCC) lattice across all concentration ratios. Standard nickel alloy monel (typically composed of roughly 67% Ni and 30% Cu) retains this uniform microstructure even after extensive cold working or thermal cycling.
This single-phase stability is the primary reason why it resists micro-galvanic corrosion. In multiphase alloys, precipitates at the grain boundaries often act as anodes or cathodes relative to the surrounding matrix, accelerating localized attack in seawater. By maintaining a homogeneous atomic structure, the alloy deprives the corrosive medium of these electrochemical trigger points.

Passivation Dynamics in Halide and Acidic Media
While many marine-grade metals rely heavily on molybdenum to resist pitting, nickel alloy monel takes a different electrochemical path. In aerated seawater, it forms a complex, tenacious protective film consisting of both nickel and copper oxides. This film is notably less brittle than standard passive layers and exhibits rapid repassivation kinetics if mechanically scratched.
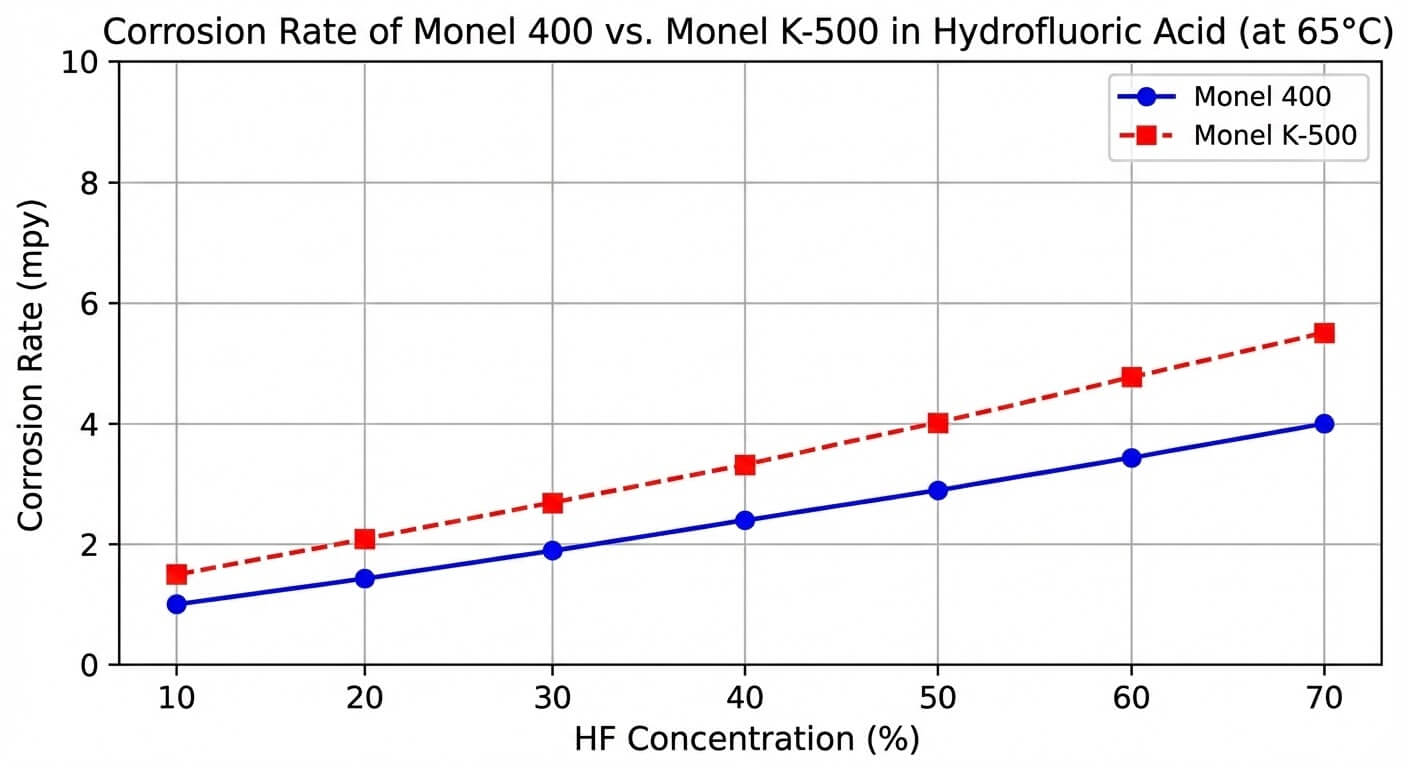
However, its performance in hydrofluoric (HF) acid is where the engineering data truly stands out. In reducing environments where stainless steels rapidly dissolve, nickel alloy monel maintains a corrosion rate of less than 1 mil per year (mpy) at room temperature. The lack of dissolved oxygen in these closed systems actually enhances its thermodynamic stability. Engineers must note, however, that introducing aeration into an HF system will significantly accelerate the anodic dissolution of the copper content, compromising the matrix.
| Parameter | Monel 400 (Solid-Solution, Annealed) | Monel K-500 (Precipitation-Hardened, Aged) |
| Nickel (Ni) | 63.0% min | 63.0% min |
| Copper (Cu) | 28.0 – 34.0% | 27.0 – 33.0% |
| Iron (Fe) | 2.5% max | 2.0% max |
| Manganese (Mn) | 2.0% max | 1.5% max |
| Carbon (C) | 0.3% max | 0.25% max |
| Al & Ti (Hardening Agents) | Not Specified |
Al: 2.30 – 3.15%
Ti: 0.35 – 0.85% |
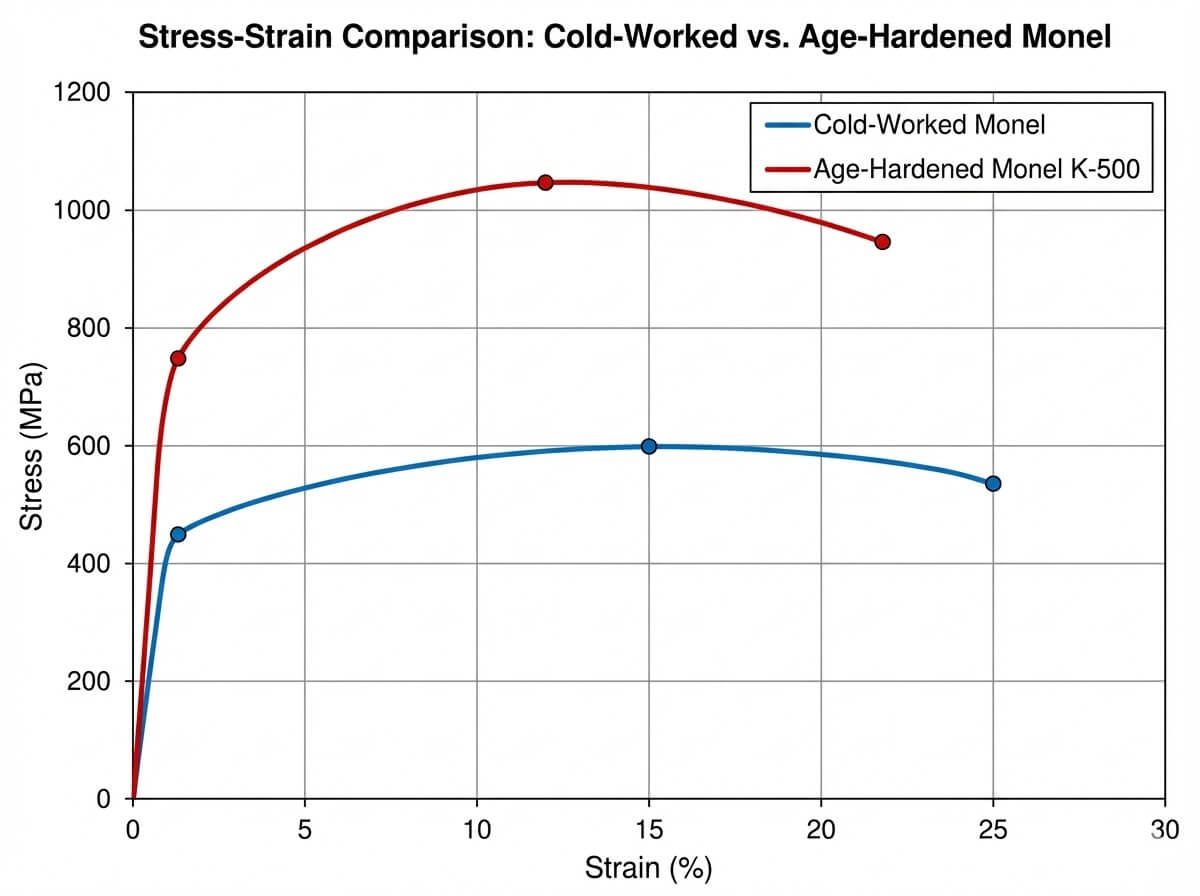
| Tensile Strength (MPa) | 480 – 550 MPa | 965 – 1100 MPa |
| Yield Strength (MPa) | 170 – 240 MPa | 690 – 790 MPa |
| Elongation (%) | 35 – 50% | 20 – 30% |
Yield Strength and Precipitation Hardening Variables
A common engineering hurdle is that solid-solution alloys often lack the high yield strength required for heavy dynamic loads, such as pump shafts or drill collars. Standard nickel alloy monel offers a baseline yield strength of around 240 MPa in the annealed condition, which can be increased via cold working but remains insufficient for high-torque applications.
To solve this, metallurgists introduced small additions of titanium and aluminum to create an age-hardenable variant. During a specific heat treatment cycle, these elements precipitate out of the solid solution as microscopic gamma-prime (γ’) particles throughout the matrix. This mechanism pins the dislocations within the crystal lattice, drastically increasing the yield strength to over 690 MPa. Crucially, this high-strength nickel alloy monel retains its exceptional dimensional stability and remains completely non-magnetic down to cryogenic temperatures—a mandatory specification for electronic sensor housings in directional drilling.
Preventing Catastrophic Failure Through Material Selection
Specifying the right grade requires a deep understanding of the exact environmental variables—specifically temperature, aeration, and flow velocity. Stagnant seawater, for instance, can induce pitting due to biofouling, whereas high-velocity brackish water leverages the alloy’s true erosion-corrosion resistance. Avoiding premature component failure means precisely matching the metallurgical state to the operational stress loads.
At 28Nickel, our engineering team routinely analyzes fluid dynamics and stress requirements to recommend the exact phase composition required for your specific application. If your current system is experiencing unexplained localized pitting, yield deformation, or fatigue, reach out to our materials engineering team for a comprehensive failure analysis and technical consultation.
Related Q&A:
1. Why does aeration negatively impact nickel alloy monel in acid applications?
In reducing acids like HF or HCl, the alloy operates near thermodynamic immunity. Aeration introduces oxygen, which shifts the oxidation-reduction potential and acts as a cathodic depolarizer. This forces the copper within the matrix into an active state, rapidly increasing the overall corrosion rate.
2. How does the gamma-prime precipitation in age-hardened Monel variants affect its machinability?
The precipitation of γ’ particles significantly increases hardness (often up to 300 HB), which accelerates tool wear. Engineers typically machine the alloy in its annealed state to near-net shape, and then perform the age-hardening heat treatment. Only final tight-tolerance grinding is executed post-aging.
3. Is nickel alloy monel susceptible to liquid metal embrittlement?
Yes. Exposure to molten metals such as mercury, lead, or bismuth can cause rapid intergranular cracking. It is critical to ensure that components manufactured from nickel alloy monel do not come into contact with these low-melting-point elements, particularly when the component is under tensile stress.